Food Chemistry Previous Exam II
University of Kentucky, Department of Animal & Food Sciences, FSC 434
1. True (T) or False (F) (2 pts. Each)
C18:2 is another way of naming linoleic acid.
The predominate sugar component of glycolipids is glucose.
Peanut oil contains a low level of cholesterol.
The melting point of a C18:1 fatty acids with cis double bounds will be lower than C18:1 fatty acids with trans double bounds.
Butyric acid is soluble in water.
Lauric acid (C12:0) is the predominant fatty acid in coconut oil.
Caseins have a very high stability to denaturation.
Proteins with less than 31.5 percent apolar amino acids usually form transparent, reversible gels.
Wheat gluten is soluble in water.
Succinylation of lysine removes its positive charge at pH 7.
BHT and BHA are better than TBHQ as antioxidants for use in bulk oil storage.
2. Draw a cis double bond. (3 pts.)
3. Draw the orientation of two fatty acids in a non-polar solvent (3 pts).
4. In general terms, describe how the hydrogenation process can result in the formation of a trans double bond (4 pts).
5. Why is hydrogen abstraction from alpha carbons preferred? (3 pts)
6. In detail, show each step of the formation of the most likely hydroperoxides from the metal catalyzed oxidation of linoleic acid. (12 pts).
7. On which carbons of linoleic acid are the hydroperoxides likely to be formed if the oxidation is initiated by singlet oxygen. (4 pts).
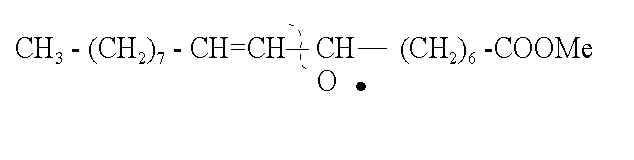
8. What two classes of compounds are most likely to be formed by the following cleavage and subsequent bond reorganization (4 pts)..

9. In twenty (20) words or less describe why a typical globular protein folds into its native tertiary structure (4 pts).
10. What three types of bonds typically stabilize the tertiary structure of proteins (3 pts).
11. Why are proteins typically denatured at pH extremes? (3 pts).
12. What type of overall charge do most proteins have at pH 7? Why?(4 pts).
13. List three ways to denature a protein besides heat and pH extremes. (3 pts).