Kentucky Pest News Newsletter
HIGHLIGHTS IN THIS ISSUE
Number 919__________ June 4, 2001
TOBACCO
ALFALFA
CORN
WHEAT
VEGETABLES
LAWN AND TURF
SHADE TREES AND ORNAMENTALS
ANNOUNCEMENTS
DIAGNOSTIC LAB HIGHLIGHTS
IPM TRAP COUNTS
ANNOUNCEMENTS
TRUE ARMYWORMS MAKE ANOTHER APPEARANCE
By Doug Johnson
 Second generation moth flight of the true armyworm has begun at the REC in Princeton, KY.
The weekly trap catch of moths
on Friday May 25 was 68 moths / trap / week, up from 7 on Friday May 18.
Already on May 29 there are about 150 moths in the trap. A daily catch rate of this size could produce a weekly trap
count of 262 moths / trap / week this Friday (6/1). This number is not quite to the first generation maximum of
398 moths / trap / week counted on Apr. 13. So what does this tell us?
Second generation moth flight of the true armyworm has begun at the REC in Princeton, KY.
The weekly trap catch of moths
on Friday May 25 was 68 moths / trap / week, up from 7 on Friday May 18.
Already on May 29 there are about 150 moths in the trap. A daily catch rate of this size could produce a weekly trap
count of 262 moths / trap / week this Friday (6/1). This number is not quite to the first generation maximum of
398 moths / trap / week counted on Apr. 13. So what does this tell us?
There are several important points to remember.
- This data represents just one location in Kentucky.
- Insect development rate is almost completely dependent upon temperature.
- So, moth flight in other locations will begin on different dates than at Princeton, depending upon temperature and
the timing of the first generation.
- Moth flight is just an indication of the beginning of the generation.
- It is not the moths that cause damage it is the caterpillars (worms). Before worm feeding begins the moths must
mate, and lay eggs; the eggs must develop and hatch. All of these processes are dependent upon temperature.
- The lower limit of temperature that will support armyworm development is about 50º F
- Most of our daily temperatures should be above 50º F during the second and subsequent generations.
- The upper limit of temperature that will support armyworm development is about 85º F.
- Above this temperature armyworms should suffer increased mortality, and reduced development.
We can use degree days to estimate how long after a moth flight occurs it will be before the worms show
up and start to feed. However, degree days are only estimates. Additionally, they deal with the
distribution through time of an insect population's development, so you will not get just a single date for an event.
If we look at the date of the peak moth flight of the first generation (Apr. 13, 2001) and the general dates we observed
massive worm feeding (May 7 to 20, 2001) we find that peak feeding occurred about three to four weeks later. Even
though this is when the infestations were noticed, I can assure you that the worms were in the field and eating at least
a week before this, so when thinking about when to go looking for second generation worms, two to three weeks after
moth flight is more appropriate.
However, insects are driven by temperature not by calendar dates. What is important is the amount of
heat units, termed degree days (DD) that accumulated during that time. From the literature we find that this development
of a complete generation should be about 1035 DD's. The DD total from Apr. 13 (peak1st Generation) to May 28
(beginning 2nd Generation) is 769 so we are about right on schedule.
The number of DD from Peak moth flight to Peak worm appearance should be about 239 DD and the
number of DD from Peak moth flight to Peak pupation (worm disappearance) should be about 739. In Princeton the two
weeks of maximum worm activity (May 7 - 20) were about 362 to 641 degree days from the peak moth flight. Again, about
the right timing. Although most people did not observe many worms before May 7, they were most certainly there.
This is borne out by the number of individuals who did not find the problem until after the damage was done.
Likewise, though problems tapered off there, were worms present after May 20th. Although not as exact as we would like,
DD's do provide us with some estimate of when the next set of worms might appear.
Now where does that leave us? We know that second generation moth flight is beginning. We know that temperatures will be
warmer from the time of this second generation moth flight to the appearance of the worms than they were for the first
generation. We don't yet know when moth flight will peak or what the daily temperatures will be for the next month.
If you use only calendar dates we should begin to look about two weeks from now.
If we begin the DD counter with last Friday, May 25th (when we noted the first increase in moth trap
catch) we are currently at 66 DD from the beginning of the moth flight. If we use temperatures from
2000 as a model for future temperatures then the first worms from the earliest moths should appear
about June 4th (235 DD from May 25th). This would be the earliest of the early worms and would likely
be very tiny, few in number, hard to find, and do little damage.
A more useful number will be the worm hatch from the peak moth flight. The peak moth flight is likely
to occur this week or next week. Again using 2000 temperatures the early hatching worms from June 1st or June 8th
should be present about June 11 or June 16th. So, it appears that we should begin scouting for worm pressure about the
end of the first week of June.
These estimates only concern WHEN worms will appear not IF or WHERE they will appear. So, the
estimate simply tells us when to go look for the pest NOT if the pests will be there.
True armyworm in Kentucky generally has three to four generations per year this is not new.
Additionally, it is not at all unknown to have spring (first generation) worm populations large enough to
require control. Generally, these cases are in small grain fields and corn planted into small grains or
fields containing grass weeds. However, before this year it has been virtually unknown for second and
subsequent generations of true armyworms to cause problems in crops. But, so far as I know we have
never had a first generation outbreak as large as the one we have seen this year. So, we best keep our
eyes open.
So, what is to be done?
- First, be aware that a second generation of true armyworm is now developing.
- Second, be prepared to go to your fields of grass crops about the second week of June and look for this insect,
- Third, be prepared to make an insecticide application if necessary.
- For forage / cattle producers this will include:
- understanding what products are available,
- Knowing the restrictions for application on or around cattle,
- Knowing time delay required before returning cattle to treated fields,
- Knowing the allowance to harvest hay from the treated fields. Forage and cattle producers should read the
Announcements section of this Kentucky Pest News to see information on the new state local need and emergency
pesticide labels.
- Producers of corn and small grains will have fewer problems with product restrictions, and more products from which
to choose.
References:
Phenology Model Database - Armyworm. Univ. CA. Statewide Integrated Pest Management Project.
http://www.ipm/ucdavis.edu/PHENOLOGY/armyworm.html
To see recent armyworm trap counts,
click here.
NEW INSECTICIDES FOR ARMYWORM CONTROL IN PASTURES
By Lee Townsend
(Frankfort) The U.S. Environmental Protection Agency has granted special registration for two insecticides to combat armyworms in pastures and hayfields in Kentucky, the Ky Department of Agriculture has announced.
The insecticides are Pounce 3.2 EC (FMC Corp.) and Confirm 2F (Rhom and Haas Co.). Both are Restricted Use pesticides. Cattle can be present during application, if necessary, when Pounce is applied and they may graze fields after treatment. However, pastures sprayed with Pounce cannot be cut for hay during the remainder of the season. Pastures sprayed with Confirm 2F can be harvested for hay after a14-day wait.
Applicators must have a copy of the appropriate state (24-c) label for Pounce or the Crisis Exemption label (Section 18) for Confirm. These are available from the pesticide dealer when the products are purchased.
WORKSHOP ON MICROSCOPIC IDENTIFICATION OF GRAY LEAF SPOT OF PERENNIAL RYEGRASS
By Paul Vincelli
Most readers who work with perennial ryegrass are
aware of the emergence of gray leaf spot in the past
decade as one of the most destructive turfgrass
diseases known. Those of you with experience
against gray leaf spot know how rapid and
destructive the disease can be.
Fungicides remain our first line of defense against
gray leaf spot, because all commercial varieties are
susceptible and cultural practices can provide only
limited control under high disease pressure.
Because of the high cost of fungicides, many turf
managers are interested in ways to use these
important products more wisely and economically.
My opinion is that at this time, there is no better
way to optimize the use of fungicides than to scout
your own swards for the disease and to
microscopically verify the presence of the causal
fungus in diseased tissues. Experienced turfgrass
pathologists agree that microscopic verification is
important because it is often not possible to positively
identify this disease based on symptoms alone.
Proper scouting can help you decide:
- when to spray
- when to switch to more costly, but more effective, fungicides
- when you can cut back on spraying
- whether to treat roughs
The University of Kentucky Cooperative Extension
Service will hold a workshop for turf managers and
other interested professionals on microscopic
identification of gray leaf spot of perennial ryegrass.
Details are as follows:
Date & Time: June 20, 2001, from 1:00-5:00
Location: The University of Kentucky campus in
Lexington
Cost: $900, Registration must be received by
Monday, May 7
While the cost may seem high at first glance, registrants
will actually be getting a lot for their money. Each
registrant will receive a high-quality compound binocular
light microscope (40-1000X magnifications, with halogen
illumination and high contrast lenses), which you will
learn to use during the workshop. In addition, each
registrant will receive dissecting kits, lense cleaning
supplies and other laboratory materials, a notebook
including color images, a 2X desk magnifier with built-in
illumination, and a field magnifying glass.
Learning Objective: As a result of this workshop,
you will be able to detect Pyricularia grisea
(the causal fungus of gray leaf spot) in diseased
turfgrass tissue. This skill will provide the
foundation for a scouting and monitoring program
for gray leaf spot.
If interested in attending, please contact Pat Yancey
at
pyancey@pop.uky.edu
pyancey@pop.uky.edu for a registration form.
Registration is limited in order to provide a high-
quality learning experience for those who attend.
Interested Kentuckians should act soon, since out-
of-state registrants will be accepted on April 20.
Registration and payment must be received by
Monday, May 7.

TOBACCO
CURRENT BLUE MOLD STATUS
By William Nesmith
 Situation:
Active blue mold is occurring in Kentucky,
Virginia, North Carolina, Georgia, Florida and
Pennsylvania. Pathogen movement via wind borne
spores and infected transplants are involved both
within states and between states. The situation is
further complicated in that the disease may be active
in communities where it is going unreported,
especially where commercial transplants may be
involved. I strongly urge the tobacco community to
work together and promptly report the status of blue
mold within the community. "Hidden" blue mold is a
major threat to the industry, especially under current
weather situations and while transplanting season is
still going. The weather situations of the past week
and that forecasted for this week should favor rapid
build up in greenhouse and transplant sites, as well as
increased activity in the field and outdoor plant beds.
Situation:
Active blue mold is occurring in Kentucky,
Virginia, North Carolina, Georgia, Florida and
Pennsylvania. Pathogen movement via wind borne
spores and infected transplants are involved both
within states and between states. The situation is
further complicated in that the disease may be active
in communities where it is going unreported,
especially where commercial transplants may be
involved. I strongly urge the tobacco community to
work together and promptly report the status of blue
mold within the community. "Hidden" blue mold is a
major threat to the industry, especially under current
weather situations and while transplanting season is
still going. The weather situations of the past week
and that forecasted for this week should favor rapid
build up in greenhouse and transplant sites, as well as
increased activity in the field and outdoor plant beds.
Shortly after Kentucky Pest News went to press last
week, we learned about active blue mold in Clark
County, Kentucky. That is why the Kentucky Blue
Mold Warning System and Ky Pest News articles
were not giving the same details. The website can be
updated daily if needed, so it should be used for the
most current status reports - it can be accessed from
the link at the end of this article.
Kentucky: Blue mold activity has been confirmed in
western Clark and eastern Fayette counties of
Kentucky, and may have spread elsewhere. These
outbreaks were confirmed on May 29, within 24 hours
of being detected by growers, and a warning was
posted for those two counties, and watches placed on
neighboring counties. Today, I am expanding the
watch, which includes the Bluegrass, Licking River,
and Northeast Kentucky Extension Areas, plus
selected counties in the Ft Harrod Area of Kentucky.
In addition, Southern Ohio and Western West Virginia
production areas should be considered under a watch.
Controls: Transplants: Statewide, preventive
fungicide spray programs should remain in place at
all tobacco transplant production sites - greenhouses,
float beds, plant beds, and distribution/holding sites.
Be sure to apply the material with sufficient pressure
to obtain complete coverage of the foliage and often
enough to keep new growth protected. See issue 908
of Kentucky Pest News,
March 12, 2001
for more specifics on chemical
options for disease control in tobacco transplant
production. Also, operate the production system as
dry as can be tolerated and still have healthy
transplants. Promptly destroy all transplants once the
transplanting season is over, but keep them protected
with fungicides until they are destroyed. We should
have adequate supplies of healthy transplants, so do
not set blue mold infected transplants; but if you feel
you must, at least alert your neighbors to what you
have done so they can increase their blue mold control
efforts.
Field: Weekly fungicide sprays should be put in place
immediately in counties under a watch or warning.
Plus, growers statewide should be scouting regularly
and become prepared to spray in a short order should
they find the disease or a watch or warning be issued
for their community. In addition, growers should be
incorporating cultural controls. If you cannot remain
informed, then start your Acrobat MZ spray program
now, or the Actigard program where plants are large
enough. See issue 910,
April 2, 2001
, Ky Pest News
for those guidelines and the fungicide
options. Also, see issue 913,
April 23, 2001
, Ky Pest
News
for information on Actigard for
blue mold prevention. Some of our crop is
approaching Actigard-treatment size, but much is too
small for this treatment now. Correctly used,
Actigard can be a valuable tool, but when poorly used
it can cause problems. Growers will need to use
Acrobat MZ until the plants are large enough to be
activated, and in some cases also return with Acrobat
MZ later in the season. The Actigard label gives the
rate at 0.5 oz/acre, starting when plants are 18 inches
tall, with a limit of two (2) applications per
crop/season - so use them wisely!
For the latest blue mold status and other tobacco disease information, check the KY Blue Mold Warning System.
 http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm
http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm
EARLY SEASON TOBACCO INSECT PESTS
By Lee Townsend
 SEASON TOBACCO INSECT TREATMENT GUIDELINES
by Lee Townsend
Insect pressure has been intense on many crops so far
this season. The same may hold true for tobacco so
careful field inspections should be made to detect
problems before serious injury has occurred. Inspect 5
groups of 20 plants in representative areas of each
field that is ½ acre or less. Add 2 additional sites for a
1 acre field and a minimum of 2 more for each
additional acre after that. Under this method there
would be 7 spots checked in a 1 acre field and 9 in a
two acre field. Avoid the ends and edge rows, these
will give you a misleading picture of insect activity in
the whole field.
SEASON TOBACCO INSECT TREATMENT GUIDELINES
by Lee Townsend
Insect pressure has been intense on many crops so far
this season. The same may hold true for tobacco so
careful field inspections should be made to detect
problems before serious injury has occurred. Inspect 5
groups of 20 plants in representative areas of each
field that is ½ acre or less. Add 2 additional sites for a
1 acre field and a minimum of 2 more for each
additional acre after that. Under this method there
would be 7 spots checked in a 1 acre field and 9 in a
two acre field. Avoid the ends and edge rows, these
will give you a misleading picture of insect activity in
the whole field.
Check wilted plants for signs of cutworm damage to
the underground portion of the stem. Examine the
bud area for aphids and budworms and the entire
plant for hornworms. Record the numbers you find
and compare them to the table below.
| Insect |
Treatment Guideline |
Weeks after transplant |
| Cutworms |
5 cut plants / 100 examined, cutworms present |
Transplant to 4 weeks after |
| Flea beetles |
3+ beetles per plants in the field less than 2 weeks, 10+ per plant on 2 to 4 week old plants that are growing rapidly |
Transplant to 4 weeks after |
| Aphids |
Colonies of 30 or more aphids on 20% of plants examined |
4 weeks after until topping |
| Budworms |
5 or more live budworms per 50 plants |
3 weeks after until button stage |
| Hornworms |
5 or more live hornworms per 50 plants |
4 weeks after until harvest |

 Tobacco flea beetles can be found on most all plants.
They chew small, round holes in the leaves. Moderate
to heavy feeding damage can result in slow initial
growth and development, especially during poor
growing conditions.
Tobacco flea beetles can be found on most all plants.
They chew small, round holes in the leaves. Moderate
to heavy feeding damage can result in slow initial
growth and development, especially during poor
growing conditions.
Tobacco aphids arrive in the field as winged adults.
These dark-bodied aphids with clear wings deposit
several live aphids on the top leaves and move to
another plant. Both green and red forms of the aphid
can be found early in the season. The green form
should be more common early in the season and the
red form more common later. Infestations may show
up first in shaded areas along fence rows.
 Tobacco budworm and hornworm moths lay single
eggs on the plant leaves. Infestations tend to be
relatively evenly spread over a field. The budworms
will be in the tightly closed bud leaves and the
pepper-like droppings may be seen there, too.
Hornworms hang from the underside of the leaf and
chew holes completely through the leaves. look on
the lower side of any leaves with rounded holes.
Tobacco budworm and hornworm moths lay single
eggs on the plant leaves. Infestations tend to be
relatively evenly spread over a field. The budworms
will be in the tightly closed bud leaves and the
pepper-like droppings may be seen there, too.
Hornworms hang from the underside of the leaf and
chew holes completely through the leaves. look on
the lower side of any leaves with rounded holes.
See
ENT-15 Insecticide Recommendations for Tobacco Beds and Fields - 2001
for the most current insecticide
alternatives.
For more information about tobacco pests, visit
"Insect Management Recommendations".
EVALUATION OF ACTIGARD 50 W AND ACROBAT MZ FOR BLUE MOLD CONTROL UNDER STRONG DISEASE PRESSURE
By William Nesmith
 Tobacco growers now have two materials labeled for
blue mold control in the field - the fungicide Acrobat
MZ and the plant activator Actigard 50 W. I have
conducted several side-by-side tests and found that
both materials will control blue mold. The Acrobat
MZ must be applied well and timely to maintain
control, however. Under strong, full-season disease
pressure, the fungicide Acrobat MZ has provided
more control than the activator at the current labeled
rates. That is clearly demonstrated in the following
data from one of last year's tests. I urge growers to
consider this as they implement their blue mold
control plan for this year.
Tobacco growers now have two materials labeled for
blue mold control in the field - the fungicide Acrobat
MZ and the plant activator Actigard 50 W. I have
conducted several side-by-side tests and found that
both materials will control blue mold. The Acrobat
MZ must be applied well and timely to maintain
control, however. Under strong, full-season disease
pressure, the fungicide Acrobat MZ has provided
more control than the activator at the current labeled
rates. That is clearly demonstrated in the following
data from one of last year's tests. I urge growers to
consider this as they implement their blue mold
control plan for this year.
Below are the data from a test conducted in the 2000
crop season at the Robinson Substation, University of
Kentucky, Quicksand, Ky. The crop was in a
Kentucky River bottom, on rotated land, and under
heavy fertilization, but according to UK's
recommendations based on soil tests. Disease
pressure was very strong, especially in August,
driven by frequent rains and fog, and it continued
strong for 10 days after topping. We were not able to
make the applications exactly on the planned dates in
several cases due to daily rains and wet soil
conditions. In hind-sight, we should have also
included a regimen that started with three Acrobat
MZ applications, then two Actigard applications, and
finished up with one or two Acrobat MZ, which this
situation warranted.
As you review these data, note the following:
- Blue mold reduced the yield by over 1200 lbs/acre.
- Blue mold was under control on Aug. 7 in all plots
receiving Acrobat MZ, but not in the check or the
one receiving Actigard alone a few days earlier.
- In plots receiving three sprays of Acrobat MZ early
in the season, followed by two applications of
Actigard, but no additional Acrobat MZ, moderate
to strong disease developed about topping time.
- The amount of systemic blue mold developing in
Actigard plots was higher than we normally
experience, which may account for the much lower
yields.
- Acrobat MZ applied at weekly intervals was the
superior treatment under these conditions, but yes,
that is a lot of spraying under difficult conditions!
|
% Leaf Area Diseased** |
|
|
|
| Treatment (Product/A) *and Planned Interval |
8/7 |
8/25 |
9/27 |
% Systemic |
Yield (lb/A) |
Crop value ($) |
| CHECK UNTREATED |
49a*** |
41a |
44a |
73ab |
1793c |
4630c |
| ACROBAT MZ (2.5 lbs/100) 7 sprays weekly-topping |
1c |
1c |
6c |
17c |
3063a |
8736a |
| ACTIGARD 50W (0.5 oz) 1st spray @ 18 inches in 4th week of growth, 2 sprays @ 10 day interval |
32b |
28b |
43a |
79a |
2134b |
5440b |
| ACROBAT MZ schedule above 1st 3 weeks, then Actigard 50W as above |
1c |
13c |
34ab |
47abc |
2245b |
5912b |
* The variety was NC-3, set June 26, topped September 5, and harvest
Sept. 27. Spray plots were single rows, 25 ft long arranged into a
randomized complete block with five replications. Full-season,
Acrobat MZ sprays were made with multiple nozzles (3 to 9)/row at
80 psi on July 13 & 19 (0.5 lbs/20 gallons), July 27 & August 7 (1.0
lbs/ 40 gallons), Aug. 15 ( 1.5 lbs/ 60 gallons) Aug. 23 (2.0 lbs/
80 gallons), and Aug 29 (2.5 lbs/ 100 gallons). The 1st Actigard
applications were made on Aug 3 with the average plants height at
19.3 and the 2nd on August 15. In all cases, the volume of material
was calculated on a per acre basis with all nozzles directed to the
plants. Additional Acrobat MZ was not applied in plots receiving
Actigard, once the Actigard applications were made.
**Disease ratings included an estimates of the % leaf surface damaged
by blue mold on various dates, plus the % of plants with systemic
stem infections taken during harvesting. Yields are the
marketable weighs and the crop value was calculated from the actual
market values obtained for this crops by grade and stalk position.
***Values within a column sharing a letter, are not significantly
different at P = 0.05 level as determined by the Tukey's hsd multiple
range test.
For the latest blue mold status and other tobacco disease information, check the KY Blue Mold Warning System.
 http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm
http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm

ALFALFA
POTATO LEAFHOPPER DAMAGE POTENTIAL
By Lee Townsend
 Potato leafhoppers, active now in Kentucky, are a key
pest of spring-seeded alfalfa and can damage the
second and third cuttings in established fields. Spring-
seeded fields are especially vulnerable to this insect
because there is plenty of time for leafhoppers to build
up to damaging numbers before the first cutting is
taken off. Ten to 14 days after cutting is the time to
check established fields for these tiny, light green
insects.
Potato leafhoppers, active now in Kentucky, are a key
pest of spring-seeded alfalfa and can damage the
second and third cuttings in established fields. Spring-
seeded fields are especially vulnerable to this insect
because there is plenty of time for leafhoppers to build
up to damaging numbers before the first cutting is
taken off. Ten to 14 days after cutting is the time to
check established fields for these tiny, light green
insects.
 Leafhoppers are sucking insects that feed on plant sap
and introduce saliva as they feed that causes "hopper
burn". This appears as a yellow, wedge-shaped area
at the tips of alfalfa leaves, and is the plant's
distinctive response to leafhopper feeding. This
damage is due to a toxic effect of the insect's saliva.
Diseases and nutritional deficiencies also cause
yellowing of alfalfa. These symptoms usually start at
the edge of the leaf but are not confined to the tips.
Intense leafhopper feeding may reduce plant height,
protein and vitamin A content of the hay. once
hopper burn is widespread in a field, significant
damage has been done. Early detection is very
important to reduce losses.
Leafhoppers are sucking insects that feed on plant sap
and introduce saliva as they feed that causes "hopper
burn". This appears as a yellow, wedge-shaped area
at the tips of alfalfa leaves, and is the plant's
distinctive response to leafhopper feeding. This
damage is due to a toxic effect of the insect's saliva.
Diseases and nutritional deficiencies also cause
yellowing of alfalfa. These symptoms usually start at
the edge of the leaf but are not confined to the tips.
Intense leafhopper feeding may reduce plant height,
protein and vitamin A content of the hay. once
hopper burn is widespread in a field, significant
damage has been done. Early detection is very
important to reduce losses.
Because of their small size, sampling of alfalfa for
potato leafhoppers requires a 15" sweep net.
Numbers of leafhoppers per sweep, along with the
height of alfalfa in the field provide the information
needed to make a control decision.
See Insect Recommendations
for more alfalfa pest recommendations.

CORN
BURCUCUMBER CONTROL--GOOD NEWS OR BAD NEWS?
By James R. Martin
 Last year was a banner season for burcucumber
problems in some areas of Kentucky. Most of the 2000
season had ample moisture for the development of
significant populations of burcucumber. In fact some
growers abandoned portions of fields and burned the
vegetation in hopes that this would kill the seed.
Last year was a banner season for burcucumber
problems in some areas of Kentucky. Most of the 2000
season had ample moisture for the development of
significant populations of burcucumber. In fact some
growers abandoned portions of fields and burned the
vegetation in hopes that this would kill the seed.
The dry weather during the early part of this season
limited the emergence of burcucumber in corn and
soybeans. This may be good news if the recent
rainfall doesn't enhance emergence of burcucumber
populations. However, if a significant amount of
burcucumber emerges as a result of the recent rainfall,
this could be bad news, particularly in cases where
corn is too large to spray a postemergence herbicide
treatment.
Examples of postemergence herbicides that will help
manage burcucumber in corn include Exceed,
Marksman, NorthStar, or Spirit for regular or
herbicide-resistant corn hybrids or Roundup
UltraMAX, Touchdown, or other glyphosate products
registered for Roundup Ready corn. Options for
soybeans include Classic for regular or herbicide-
resistant varieties; or Roundup UltraMAX,
Touchdown, or other glyphosate products registered
for Roundup Ready soybean; or Synchrony STS for
STS soybean. Information on herbicide rates and
timing of application for burcucumber control in corn
or soybean is summarized in the following tables:
| Table 1. Postemergence Herbicides For Burcucumber Control in Corn. |
| Postemergence Herbicide |
Rate/A |
Burcucumber Max. Size |
Corn Size/Stage |
| Exceed |
1 oz |
8" |
Broadcast between 4"and 30" tall.Use drop nozzles when field corn is 20" to 30" tall or exhibits more than 6
collars V6 (whichever occurs first). |
| Marksman |
3.5 pt |
3" |
Through fifth leaf stage or 8" tall (whichever occurs first). |
| NorthStar |
5 oz |
4" |
Broadcast between 4"and 20" tall.Use drop nozzles when field corn is 20" (V6) up to 36" tall or 15 days before
tassel emergence (whichever occurs first). |
| Roundup UltraMAX (RR Corn only) |
20 oz |
12" |
Through V8 stage or 30" tall (whichever occurs first). |
| 26 oz |
18" |
| Spirit |
1 oz |
6" |
Broadcast between 4"and 20" tall. Use drop nozzles when field corn is 20" to 24" tall or exhibits more than
6 collars V6 (whichever occurs first). |
| Touchdown (IQ) (RR Corn only) |
24 oz |
6" |
Through V8 stage or 30" tall (whichever occurs first). |
| 32 oz |
12" |
| Table 2. Postemergence Herbicides For Burcucumber Control in Soybeans. |
| Postemergence Herbicide |
Rate/A |
Burcucumber Max. Size |
Soybean Stage |
| Classic |
0.67 oz |
2 - 3" |
After first trifoliate has expanded and at least 60 days before maturity. |
| 0.75 |
2 - 6" |
| Extreme |
3 pt |
6" |
Before bloom and prior to 85 days of harvest. |
| RoundupUtlraMAX (RR Soybean only) |
20 oz |
12" |
From cracking through full flowering. |
| 26 oz |
18" |
| Synchrony STS (STS Soybean only) |
0.5 oz |
2 -3" |
After first trifoliate has expanded and at least 60 days before maturity. |
| Touchdown (IQ) (RR Soybean only) |
24 oz |
6" |
From cracking through full flowering. |
| 32 oz |
12" |
It is not realistic to expect season-long control of burcucumber in
corn or soybeans. Achieving early-season control is an important
step in managing burcucumber; however, the ultimate impact in terms
of harvesting corn or soybeans will likely depend on weather
conditions affecting late- season emergence and growth of this
problem weed.
For more information about corn pests, visit
"Insect Management Recommendations".

WHEAT
WHEAT DISEASE UPDATE
By Don Hershman
 As the wheat season comes to a close, I have not
moved away from my earlier comment that this year
has produced the least amount of disease of any crop I
have seen since coming to Kentucky 17 years ago.
There was a rebound in Stagonospora leaf and glume
blotch in response to some recent rain events. And, of
course, there were hits of most other diseases here and
there, but overall, diseases will have a very minor
impact on the state's wheat yields this season.
As the wheat season comes to a close, I have not
moved away from my earlier comment that this year
has produced the least amount of disease of any crop I
have seen since coming to Kentucky 17 years ago.
There was a rebound in Stagonospora leaf and glume
blotch in response to some recent rain events. And, of
course, there were hits of most other diseases here and
there, but overall, diseases will have a very minor
impact on the state's wheat yields this season.
It is my opinion that the overall low levels of disease
is the result of three factors. First, we did not have a
mild winter like we have experienced the last couple
of years. UK extension entomologist Dr. Doug
Johnson predicted in January that the cold winter
should reduce the aphid vectors that transmit barley
yellow dwarf virus. It appears as though his
prediction was accurate in that both aphids and barley
yellow dwarf were minimal. However, I cannot
explain the overall lack of the soil-borne (fungal-
transmitted) viruses; the soil conditions in the fall and
late winter seemed to be ideal for infection and
symptom expression. This situation underscores the
unpredictability inherent in many biological systems.
In addition to the cold winter, I believe that the late
spring cold snap significantly reduced fungal
populations. I have seen this phenomenon before. It
appears that our common foliar fungal pathogens are
fairly cold-sensitive and are easily killed-off in
response to the wide temperature fluctuations that
common in early spring in Kentucky. In the case of
leaf rust, which is very easily killed off by borderline
freezing temperatures, that pathogen barely made it
into the state as most of the rust inoculum was killed
off by abnormally cold weather in Texas and other
mid-south states. There simply were very low levels
of leaf rust inocula available to blow into Kentucky.
Finally, the exceptionally dry weather we experienced
throughout most of the state this spring most certainly
limited the proliferation of fungal pathogens. If you
made a fungicide application this spring, there is very
little chance that you will get a break-even return on
your investment. Unfortunately, even the best
fungicide use decisions cannot take into account
future weather patterns that may negate the value of a
fungicide application. This is one of the many inherent
risks associated with wheat production.

VEGETABLES
VEGETABLE INSECT UPDATE
By Ric Bessin
So far this season we have seen considerable pest
insect activity in a number of field and specialty crops.
Vegetable producers should be on the watch for
several early season pests, and some producers have
already reported problems with cucumber beetles and
other pests. Particularly with vegetable insect control,
early season management can be critical.
Striped Cucumber beetles
 There have been numerous reports of high cucumber
beetle activity in melons, cucumbers, and squash.
Bacterial wilt has also been reported in several areas.
Some producers have treated their crops with
systemic insecticides at planting but are noticing
beetles returning 3 to 5 weeks after treatment. There
have been some reports of poor performance with a
newly registered systemic insecticide for cucumber
beetle control. However, the poor performance was
not related to the product, rather it was used in the
wrong manner and below labeled rates. The length of
activity of these treatments will depend on the rate of
the insecticides used. Producers will need to continue
to scout these crops and use foliar treatments as
necessary when beetle activity increases.
There have been numerous reports of high cucumber
beetle activity in melons, cucumbers, and squash.
Bacterial wilt has also been reported in several areas.
Some producers have treated their crops with
systemic insecticides at planting but are noticing
beetles returning 3 to 5 weeks after treatment. There
have been some reports of poor performance with a
newly registered systemic insecticide for cucumber
beetle control. However, the poor performance was
not related to the product, rather it was used in the
wrong manner and below labeled rates. The length of
activity of these treatments will depend on the rate of
the insecticides used. Producers will need to continue
to scout these crops and use foliar treatments as
necessary when beetle activity increases.
Beet Armyworm
 A early report indicated that high numbers of beet
armyworm are active in western Tennessee. To date,
pheromone traps in Kentucky have revealed only low
activity. As beet armyworm populations will continue
to increase until the first frosts of fall, this is an insect
that we will need to continue to watch for on peppers,
tomatoes, cole crops, and sweet corn. Our trapping
numbers has been close to one to two moths per week
per trap. Generally, we do not begin to see problems
in vegetable crops until number approach 20 to 30 per
trap per week. Beet armyworm commonly attacks
pigweed, so it is a good idea to occasionally scout
pigweed for signs of beet armyworm. If beet
armyworm does become a problem, early control and
proper selection of insecticides are critical to
acceptable control.
A early report indicated that high numbers of beet
armyworm are active in western Tennessee. To date,
pheromone traps in Kentucky have revealed only low
activity. As beet armyworm populations will continue
to increase until the first frosts of fall, this is an insect
that we will need to continue to watch for on peppers,
tomatoes, cole crops, and sweet corn. Our trapping
numbers has been close to one to two moths per week
per trap. Generally, we do not begin to see problems
in vegetable crops until number approach 20 to 30 per
trap per week. Beet armyworm commonly attacks
pigweed, so it is a good idea to occasionally scout
pigweed for signs of beet armyworm. If beet
armyworm does become a problem, early control and
proper selection of insecticides are critical to
acceptable control.
Squash Bug
 Producers of summer and winter squash varieties
should be on the watch for squash bugs. They
overwinter as adults and begin to move into fields in
early June. Females lay their eggs on the undersides of
leaves in the angles between major leaf veins. Eggs a
small, football shaped, bronze in color, and laid in
groups of 10 to 20. The threshold for squash bugs is an
average of one egg cluster per plant. The best time to
control squash bug is just after the eggs hatch. Early
control is important as delaying application means
that the foliage will be denser, interfering with
coverage. Also, smaller, younger squash bug nymphs
are less difficult to control.
Producers of summer and winter squash varieties
should be on the watch for squash bugs. They
overwinter as adults and begin to move into fields in
early June. Females lay their eggs on the undersides of
leaves in the angles between major leaf veins. Eggs a
small, football shaped, bronze in color, and laid in
groups of 10 to 20. The threshold for squash bugs is an
average of one egg cluster per plant. The best time to
control squash bug is just after the eggs hatch. Early
control is important as delaying application means
that the foliage will be denser, interfering with
coverage. Also, smaller, younger squash bug nymphs
are less difficult to control.
European Corn Borer
 The first generation of the European corn borer was
early and strong this spring. In some respects, it may
have been out of synchrony with the corn as corn has
been slow to grow in some areas. With high moth
numbers and small corn, it is not uncommon to find
some egg laying on other vegetables such as tomatoes,
peppers, and potatoes. European corn borers
occasionally bores into the stems of these plants when
fruit are not available. The early start this spring may
mean that we are likely to experience attack by three
European corn borer generations this year instead of
two.
The first generation of the European corn borer was
early and strong this spring. In some respects, it may
have been out of synchrony with the corn as corn has
been slow to grow in some areas. With high moth
numbers and small corn, it is not uncommon to find
some egg laying on other vegetables such as tomatoes,
peppers, and potatoes. European corn borers
occasionally bores into the stems of these plants when
fruit are not available. The early start this spring may
mean that we are likely to experience attack by three
European corn borer generations this year instead of
two.
Squash Vine Borer
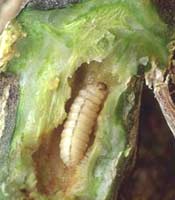 The squash vine borer is a key pest of winter squash,
gourds and pumpkins in Kentucky. Unfortunately, it
is usually noticed only after it has done its damage.
Symptoms appear in mid-summer when a long
runner or an entire plant wilts suddenly. Adult moths
begin to emerge about the time the plants begin to
run, and moth flight continues through mid August.
Only the unmated adult bugs overwinter in Kentucky.
The key to squash vine borer management is
controlling the borers before they enter the stem. Once
inside the vine, insecticidal control is ineffective. Poor
timing of sprays is the usual cause of inadequate
control. Monitor plants weekly from mid-June
through August for initial signs of the borer's frass at
entrance holes in the stems. Very early signs of larval
feeding indicate that other eggs will be hatching soon.
Use two insecticide applications 7 days apart to
control The squash vine borer is a key pest of winter
squash, gourds and pumpkins in Kentucky.
Unfortunately, it is hatching larvae and continue to
monitor for additional activity. Sprays need to
penetrate the canopy to cover the vines to be effective.
The squash vine borer is a key pest of winter squash,
gourds and pumpkins in Kentucky. Unfortunately, it
is usually noticed only after it has done its damage.
Symptoms appear in mid-summer when a long
runner or an entire plant wilts suddenly. Adult moths
begin to emerge about the time the plants begin to
run, and moth flight continues through mid August.
Only the unmated adult bugs overwinter in Kentucky.
The key to squash vine borer management is
controlling the borers before they enter the stem. Once
inside the vine, insecticidal control is ineffective. Poor
timing of sprays is the usual cause of inadequate
control. Monitor plants weekly from mid-June
through August for initial signs of the borer's frass at
entrance holes in the stems. Very early signs of larval
feeding indicate that other eggs will be hatching soon.
Use two insecticide applications 7 days apart to
control The squash vine borer is a key pest of winter
squash, gourds and pumpkins in Kentucky.
Unfortunately, it is hatching larvae and continue to
monitor for additional activity. Sprays need to
penetrate the canopy to cover the vines to be effective.

LAWN & TURF
RESISTANCE TO QOI (=STROBILURIN-LIKE) FUNGICIDES IN GRAY LEAF SPOT OF PERENNIAL RYEGRASS
By Paul Vincelli
Gray leaf spot, caused by the fungus Pyricularia grisea,
is the most destructive disease of perennial ryegrass.
In Kentucky, the disease usually first occurs in early
to mid-August, and can develop very rapidly to
destructive levels anytime through September.
Cultural practices such as minimizing N fertility can
reduce disease activity. However, perennial ryegrass
is so susceptible that fungicide must be applied
during periods of high disease pressure.
Among the available products, the most effective
fungicides are thiophanate methyl and the QoI
fungicides (the QoI fungicides were previously called
"strobilurins", but the name has been changed since
there are strobilurin fungicides as well as non-
strobilurin fungicides which have the very same mode
of action). QoI fungicides available for gray leaf spot
control include Heritage (active
ingredient=azoxystrobin) and Compass
(trifloxystrobin). Unfortunately, there is a significant
risk for the development of resistant strains to the QoI
fungicides and thiophanate methyl, especially given
the highly adaptable nature of P. grisea.
Occurrence of QoI-Resistant Strains
Resistant strains of P. grisea from perennial ryegrass
have emerged rather quickly, given that QoI
fungicides have only been labeled against this disease
since 1998. Three golf courses--a course in Lexington
and two in Illinois experienced significant damage
from the disease last year on fairways treated with
Heritage (areas affected ranged from 2.7 to 22 acres).
At the course in Lexington, we had a research trial
located in a fairway where the disease developed, so
we have a good data set on the progress of that
outbreak. All three courses had a history of using
Heritage on fairways; fairways where outbreaks
occurred had been treated a total of 5-11 times since
1998.
Fungal isolates of P. grisea were collected from all
three courses (thanks to Dr. Andy Hamblin for
providing isolates from the courses in Illinois). These
"suspect isolates" as well as a set of "baseline isolates"
never exposed to QoI fungicides were tested for
sensitivity to QoI fungicides in tests in petri diseases
as well as on perennial ryegrass. The tests with
perennial ryegrass show partial to complete loss of
control with Heritage and, for three of four isolates,
with Compass (Table 1). Our experiments in petri
dishes were extensive, with repeated tests of
sensitivity of 21 isolates to three fungicides at
numerous concentrations. In those tests, with one
exception, suspect isolates were found to be 700-800
times less sensitive to QoI fungicides than baseline
isolates. Molecular genetics research in the laboratory
of Dr. Mark Farman (in the UK Dept. of Plant
Pathology) revealed that all suspect isolates tested
possessed one of two mutations known to be
associated with QoI resistance.
The data in Table 1 also show cross-resistance: strains
resistant to Heritage were also resistant to Compass,
even though the latter had never been used on those
fairways. This is what one would expect for
fungicides that share a common mode of action.
Implications for Management
The rather rapid development of Qo I-resistant
subpopulations of P. grisea strains from perennial
ryegrass suggests that the potential for Qo I resistance
in this pathogen is significant. However, rapid
development of a resistant subpopulation in the field
does not necessarily preclude the effective use of the
fungicide in other locations for a number of years. For
example, resistance in the cucurbit powdery mildew
fungus to benomyl and to DMI fungicides occurred 1
and 2 years after commercial introduction,
respectively, and yet these fungicides remained
important disease control tools in other areas for years
until resistant strains became widespread.
In my opinion, the QoI fungicides will continue to
have an important place for gray leaf spot control; this
report does not change that fact. Furthermore, it
would be unwise for superintendents to avoid the use
of QoI fungicides for gray leaf spot control, locations
where resistance has not been detected. Taking
Heritage and Compass out of the spray program puts
an awful lot of selection pressure for resistance to
thiophanate-methyl, the only remaining "four-star"
product. I have the pessimistic belief that we will
eventually discover resistance in ryegrass-infecting
strains of P. grisea to thiophanate-methyl, perhaps
soon. Thus, superintendents should do all they can to
minimize that risk by keeping QoI fungicides in their
spray program until such time as these products no
longer are effective on their course. See below for
specific recommendations.
Furthermore, given the significant risk of resistance
development to QoI fungicides, my advice is to scout
your courses regularly after using QoI fungicides,
should application of alternative products be
necessary.
Practices for Minimizing Resistance Risk
Use of the following practices will reduce -- but not
eliminate -- the risk of resistance to QoI fungicides.
- Minimize disease pressure through cultural
practices to the extent possible. This will reduce the
chance of a resistant mutant by reducing the size of
the pathogen population. If fertilizing during the
period from June through August, foliar feed with no
more than 0.1 to 0.25 lb soluble nitrogen/1000 ft2
during that period. Schedule irrigations near sunrise
and avoid evening irrigation.
- Minimize consecutive applications of fungicides
having a similar mode of action, especially during the
period of highest disease pressure. In fact, my
recommendation has been to switch to a different
mode of action each time you spray for gray leaf spot.
- Tank-mix products having different modes of action
during periods of high disease pressure.
Chlorothalonil, a multi-site inhibitor, is an especially
good mixing partner from the standpoint of resistance
management. There is essentially no significant risk
of resistance to a multi-site inhibitor fungicide. This is
an especially important mixing partner for curative
situations.
- Switching from the QoI fungicides and thiophanate
methyl as disease pressure diminishes, because this
reduces the exposure of P. grisea to these at-risk
fungicides.
| Table 1. Sensitivity of isolates of P. grisea to QoI fungicides on perennial ryegrass. |
|
% Foliar Blighting |
|
Baseline isolates |
Resistant isolates |
| Treatment |
KY-96 |
MS-97 |
OH-97 |
CHRF |
RGNJ |
Il-00-1 |
GG-5-2b-2 |
GG-2b |
HR2000-2-2 |
| Water |
58.5 a |
70.5 a |
67.5 a |
44 a |
67.3 a |
62.0 a |
79.5 a |
80.8 a |
14.3 a |
| Heritage |
0 b |
1.4 b |
0 b |
0 b |
0 b |
18.5 b |
62.5 a |
57.8 b |
5.5 b |
| Compass |
0 b |
0 b |
0 b |
0 b |
0 b |
0.3 c |
69.5 a |
72.5 ab |
13.5 a |
Means within a column followed by the same letter are not significantly different, Tukey's studentized range test, P = 0.05.

SHADE TREES AND ORNAMENTALS
LEAF SPOTS CAN DAMAGE ENGLISH IVY
By John Hartman
In early spring, winter injury was noted in many ivy
beds. Summer weather brings on additional
problems, primarily leaf spot diseases. There are two
important English Ivy (Hedera) leaf spot diseases in
Kentucky, one caused by a bacterium, and one by a
fungus. The two diseases are sometimes difficult to
distinguish.
Bacterial leaf spot is favored by periods of warm, wet
weather typical of summer in Kentucky. This disease,
caused by the bacterium Xanthomonas campestris pv.
hederae, can be especially damaging to Ivy growing in
many landscapes. The bacteria invade leaves, shoots,
and stems through stomata and wounds causing a
greenish-brown angular leaf spot 1/4 to 1/2 inch or
larger in size. The spots sometimes appear greasy and
may have a yellow margin; as they age, spots turn
dark brown and may crack as they dry.
The disease is diagnosed in the laboratory by
observing bacterial streaming under the microscope;
however, the disease is often so active that county
agents or landscape industry specialists can also
diagnose the disease in the field in the same way. Cut
through several leaf spots with a sharp knife and
place small infected leaf pieces on a glass slide. Add a
drop or two of clean water to the infected tissue and
cover with small glass cover slip. After a few minutes
to an hour, bacterial streaming can be seen just by
holding the glass slide up to the light and observing
the milky color of the water near the dissected leaf
spot.
Growers should avoid planting diseased plant
material, and avoid sprinkler irrigation which
splashes bacteria from diseased to healthy plants.
Copper-based fungicides which also serve as
bactericides can be used to slow the spread of bacterial
leaf spot.
Fungal leaf spot (anthracnose) appears as large,
irregularly shaped tan or brown spots having
numerous pimple-like fungal fruiting structures in the
dead tissues. The causal fungus is Glomerella cingulata;
however, the imperfect fungal state, Colletotrichum is
normally observed now. Close examination with a
hand lens may show spine-like formations associated
with the fungal fruiting structures. There are other
fungal leaf spot diseases of English Ivy which also
produce fungal fruiting structures; this distinguishes
them from bacterial spot, which produces none.
Fungal spots do not produce bacterial streaming as
described above.
Controls for anthracnose and fungal leaf spots are
similar to controls for bacterial leaf spot, except that
fungicides such as thiophanate-methyl [Cleary's 3336]
(cleared for anthracnose of landscape plants) and
mancozeb [Fore] (used for fungal leaf spots of
ornamentals) are also available.

DIAGNOSTIC LAB HIGHLIGHTS
DIAGNOSTIC LAB - HIGHLIGHTS
By Julie Beale and Paul Bachi
Rainy weather in central and eastern Kentucky and
continued dry conditions in western Kentucky have
promoted a variety of disease and environmental
problems recently. In the Diagnostic lab we have seen
zinc and temporary phosphorus deficiency in corn;
herbicide injury, cold injury (field), temporary
phosphorus deficiency, manganese toxicity, Pythium
root rot, target spot, bacterial blackleg, tomato spotted
wilt virus and tobacco ringspot virus on tobacco.
On fruit and vegetable crops, we have seen
anthracnose on blackberry; fireblight on apple and
pear; black knot on cherry and plum; oriental fruit
moth injury on peach; Alternaria fruit rot on squash;
root knot nematode and numerous samples of
bacterial wilt on cantaloupe; and early blight, bacterial
canker and tomato spotted wilt on tomato.
On ornamentals, we have seen crown gall on
rosemary; bacterial leaf spot on ivy; black root rot on
holly; Kabatina twig blight on juniper; Pseudonectria
canker on boxwood; and lace bug injury on azalea.

IPM TRAP COUNTS:
By Patty Lucas, University of Kentucky Research Center
Insect Trap Counts
| UKREC-Princeton, KY, May 25 - June 1, 2001
|
| Black Cutworm
| 14
|
| True Armyworm
| 172
|
| Fall Armyworm
| 0
|
| Beet Armyworm
| 1
|
| Corn Earworm
| 3
|
| European Corn Borer
| 0
|
| Southwestern Corn Borer
| 19
|
To see recent moth trap counts,
click here.
Lee Townsend
Extension Entomologist
BACK
TO KY PEST NEWS HOME
 Second generation moth flight of the true armyworm has begun at the REC in Princeton, KY.
The weekly trap catch of moths
on Friday May 25 was 68 moths / trap / week, up from 7 on Friday May 18.
Already on May 29 there are about 150 moths in the trap. A daily catch rate of this size could produce a weekly trap
count of 262 moths / trap / week this Friday (6/1). This number is not quite to the first generation maximum of
398 moths / trap / week counted on Apr. 13. So what does this tell us?
Second generation moth flight of the true armyworm has begun at the REC in Princeton, KY.
The weekly trap catch of moths
on Friday May 25 was 68 moths / trap / week, up from 7 on Friday May 18.
Already on May 29 there are about 150 moths in the trap. A daily catch rate of this size could produce a weekly trap
count of 262 moths / trap / week this Friday (6/1). This number is not quite to the first generation maximum of
398 moths / trap / week counted on Apr. 13. So what does this tell us?

 Situation:
Active blue mold is occurring in Kentucky,
Virginia, North Carolina, Georgia, Florida and
Pennsylvania. Pathogen movement via wind borne
spores and infected transplants are involved both
within states and between states. The situation is
further complicated in that the disease may be active
in communities where it is going unreported,
especially where commercial transplants may be
involved. I strongly urge the tobacco community to
work together and promptly report the status of blue
mold within the community. "Hidden" blue mold is a
major threat to the industry, especially under current
weather situations and while transplanting season is
still going. The weather situations of the past week
and that forecasted for this week should favor rapid
build up in greenhouse and transplant sites, as well as
increased activity in the field and outdoor plant beds.
Situation:
Active blue mold is occurring in Kentucky,
Virginia, North Carolina, Georgia, Florida and
Pennsylvania. Pathogen movement via wind borne
spores and infected transplants are involved both
within states and between states. The situation is
further complicated in that the disease may be active
in communities where it is going unreported,
especially where commercial transplants may be
involved. I strongly urge the tobacco community to
work together and promptly report the status of blue
mold within the community. "Hidden" blue mold is a
major threat to the industry, especially under current
weather situations and while transplanting season is
still going. The weather situations of the past week
and that forecasted for this week should favor rapid
build up in greenhouse and transplant sites, as well as
increased activity in the field and outdoor plant beds.
 SEASON TOBACCO INSECT TREATMENT GUIDELINES
by Lee Townsend
Insect pressure has been intense on many crops so far
this season. The same may hold true for tobacco so
careful field inspections should be made to detect
problems before serious injury has occurred. Inspect 5
groups of 20 plants in representative areas of each
field that is ½ acre or less. Add 2 additional sites for a
1 acre field and a minimum of 2 more for each
additional acre after that. Under this method there
would be 7 spots checked in a 1 acre field and 9 in a
two acre field. Avoid the ends and edge rows, these
will give you a misleading picture of insect activity in
the whole field.
SEASON TOBACCO INSECT TREATMENT GUIDELINES
by Lee Townsend
Insect pressure has been intense on many crops so far
this season. The same may hold true for tobacco so
careful field inspections should be made to detect
problems before serious injury has occurred. Inspect 5
groups of 20 plants in representative areas of each
field that is ½ acre or less. Add 2 additional sites for a
1 acre field and a minimum of 2 more for each
additional acre after that. Under this method there
would be 7 spots checked in a 1 acre field and 9 in a
two acre field. Avoid the ends and edge rows, these
will give you a misleading picture of insect activity in
the whole field.

 Tobacco budworm and hornworm moths lay single
eggs on the plant leaves. Infestations tend to be
relatively evenly spread over a field. The budworms
will be in the tightly closed bud leaves and the
pepper-like droppings may be seen there, too.
Hornworms hang from the underside of the leaf and
chew holes completely through the leaves. look on
the lower side of any leaves with rounded holes.
Tobacco budworm and hornworm moths lay single
eggs on the plant leaves. Infestations tend to be
relatively evenly spread over a field. The budworms
will be in the tightly closed bud leaves and the
pepper-like droppings may be seen there, too.
Hornworms hang from the underside of the leaf and
chew holes completely through the leaves. look on
the lower side of any leaves with rounded holes.

 Potato leafhoppers, active now in Kentucky, are a key
pest of spring-seeded alfalfa and can damage the
second and third cuttings in established fields. Spring-
seeded fields are especially vulnerable to this insect
because there is plenty of time for leafhoppers to build
up to damaging numbers before the first cutting is
taken off. Ten to 14 days after cutting is the time to
check established fields for these tiny, light green
insects.
Potato leafhoppers, active now in Kentucky, are a key
pest of spring-seeded alfalfa and can damage the
second and third cuttings in established fields. Spring-
seeded fields are especially vulnerable to this insect
because there is plenty of time for leafhoppers to build
up to damaging numbers before the first cutting is
taken off. Ten to 14 days after cutting is the time to
check established fields for these tiny, light green
insects.
 Leafhoppers are sucking insects that feed on plant sap
and introduce saliva as they feed that causes "hopper
burn". This appears as a yellow, wedge-shaped area
at the tips of alfalfa leaves, and is the plant's
distinctive response to leafhopper feeding. This
damage is due to a toxic effect of the insect's saliva.
Diseases and nutritional deficiencies also cause
yellowing of alfalfa. These symptoms usually start at
the edge of the leaf but are not confined to the tips.
Intense leafhopper feeding may reduce plant height,
protein and vitamin A content of the hay. once
hopper burn is widespread in a field, significant
damage has been done. Early detection is very
important to reduce losses.
Leafhoppers are sucking insects that feed on plant sap
and introduce saliva as they feed that causes "hopper
burn". This appears as a yellow, wedge-shaped area
at the tips of alfalfa leaves, and is the plant's
distinctive response to leafhopper feeding. This
damage is due to a toxic effect of the insect's saliva.
Diseases and nutritional deficiencies also cause
yellowing of alfalfa. These symptoms usually start at
the edge of the leaf but are not confined to the tips.
Intense leafhopper feeding may reduce plant height,
protein and vitamin A content of the hay. once
hopper burn is widespread in a field, significant
damage has been done. Early detection is very
important to reduce losses.

 Last year was a banner season for burcucumber
problems in some areas of Kentucky. Most of the 2000
season had ample moisture for the development of
significant populations of burcucumber. In fact some
growers abandoned portions of fields and burned the
vegetation in hopes that this would kill the seed.
Last year was a banner season for burcucumber
problems in some areas of Kentucky. Most of the 2000
season had ample moisture for the development of
significant populations of burcucumber. In fact some
growers abandoned portions of fields and burned the
vegetation in hopes that this would kill the seed.

 As the wheat season comes to a close, I have not
moved away from my earlier comment that this year
has produced the least amount of disease of any crop I
have seen since coming to Kentucky 17 years ago.
There was a rebound in Stagonospora leaf and glume
blotch in response to some recent rain events. And, of
course, there were hits of most other diseases here and
there, but overall, diseases will have a very minor
impact on the state's wheat yields this season.
As the wheat season comes to a close, I have not
moved away from my earlier comment that this year
has produced the least amount of disease of any crop I
have seen since coming to Kentucky 17 years ago.
There was a rebound in Stagonospora leaf and glume
blotch in response to some recent rain events. And, of
course, there were hits of most other diseases here and
there, but overall, diseases will have a very minor
impact on the state's wheat yields this season.

 There have been numerous reports of high cucumber
beetle activity in melons, cucumbers, and squash.
Bacterial wilt has also been reported in several areas.
Some producers have treated their crops with
systemic insecticides at planting but are noticing
beetles returning 3 to 5 weeks after treatment. There
have been some reports of poor performance with a
newly registered systemic insecticide for cucumber
beetle control. However, the poor performance was
not related to the product, rather it was used in the
wrong manner and below labeled rates. The length of
activity of these treatments will depend on the rate of
the insecticides used. Producers will need to continue
to scout these crops and use foliar treatments as
necessary when beetle activity increases.
There have been numerous reports of high cucumber
beetle activity in melons, cucumbers, and squash.
Bacterial wilt has also been reported in several areas.
Some producers have treated their crops with
systemic insecticides at planting but are noticing
beetles returning 3 to 5 weeks after treatment. There
have been some reports of poor performance with a
newly registered systemic insecticide for cucumber
beetle control. However, the poor performance was
not related to the product, rather it was used in the
wrong manner and below labeled rates. The length of
activity of these treatments will depend on the rate of
the insecticides used. Producers will need to continue
to scout these crops and use foliar treatments as
necessary when beetle activity increases.
 A early report indicated that high numbers of beet
armyworm are active in western Tennessee. To date,
pheromone traps in Kentucky have revealed only low
activity. As beet armyworm populations will continue
to increase until the first frosts of fall, this is an insect
that we will need to continue to watch for on peppers,
tomatoes, cole crops, and sweet corn. Our trapping
numbers has been close to one to two moths per week
per trap. Generally, we do not begin to see problems
in vegetable crops until number approach 20 to 30 per
trap per week. Beet armyworm commonly attacks
pigweed, so it is a good idea to occasionally scout
pigweed for signs of beet armyworm. If beet
armyworm does become a problem, early control and
proper selection of insecticides are critical to
acceptable control.
A early report indicated that high numbers of beet
armyworm are active in western Tennessee. To date,
pheromone traps in Kentucky have revealed only low
activity. As beet armyworm populations will continue
to increase until the first frosts of fall, this is an insect
that we will need to continue to watch for on peppers,
tomatoes, cole crops, and sweet corn. Our trapping
numbers has been close to one to two moths per week
per trap. Generally, we do not begin to see problems
in vegetable crops until number approach 20 to 30 per
trap per week. Beet armyworm commonly attacks
pigweed, so it is a good idea to occasionally scout
pigweed for signs of beet armyworm. If beet
armyworm does become a problem, early control and
proper selection of insecticides are critical to
acceptable control.
 Producers of summer and winter squash varieties
should be on the watch for squash bugs. They
overwinter as adults and begin to move into fields in
early June. Females lay their eggs on the undersides of
leaves in the angles between major leaf veins. Eggs a
small, football shaped, bronze in color, and laid in
groups of 10 to 20. The threshold for squash bugs is an
average of one egg cluster per plant. The best time to
control squash bug is just after the eggs hatch. Early
control is important as delaying application means
that the foliage will be denser, interfering with
coverage. Also, smaller, younger squash bug nymphs
are less difficult to control.
Producers of summer and winter squash varieties
should be on the watch for squash bugs. They
overwinter as adults and begin to move into fields in
early June. Females lay their eggs on the undersides of
leaves in the angles between major leaf veins. Eggs a
small, football shaped, bronze in color, and laid in
groups of 10 to 20. The threshold for squash bugs is an
average of one egg cluster per plant. The best time to
control squash bug is just after the eggs hatch. Early
control is important as delaying application means
that the foliage will be denser, interfering with
coverage. Also, smaller, younger squash bug nymphs
are less difficult to control.
 The first generation of the European corn borer was
early and strong this spring. In some respects, it may
have been out of synchrony with the corn as corn has
been slow to grow in some areas. With high moth
numbers and small corn, it is not uncommon to find
some egg laying on other vegetables such as tomatoes,
peppers, and potatoes. European corn borers
occasionally bores into the stems of these plants when
fruit are not available. The early start this spring may
mean that we are likely to experience attack by three
European corn borer generations this year instead of
two.
The first generation of the European corn borer was
early and strong this spring. In some respects, it may
have been out of synchrony with the corn as corn has
been slow to grow in some areas. With high moth
numbers and small corn, it is not uncommon to find
some egg laying on other vegetables such as tomatoes,
peppers, and potatoes. European corn borers
occasionally bores into the stems of these plants when
fruit are not available. The early start this spring may
mean that we are likely to experience attack by three
European corn borer generations this year instead of
two.
 The squash vine borer is a key pest of winter squash,
gourds and pumpkins in Kentucky. Unfortunately, it
is usually noticed only after it has done its damage.
Symptoms appear in mid-summer when a long
runner or an entire plant wilts suddenly. Adult moths
begin to emerge about the time the plants begin to
run, and moth flight continues through mid August.
Only the unmated adult bugs overwinter in Kentucky.
The key to squash vine borer management is
controlling the borers before they enter the stem. Once
inside the vine, insecticidal control is ineffective. Poor
timing of sprays is the usual cause of inadequate
control. Monitor plants weekly from mid-June
through August for initial signs of the borer's frass at
entrance holes in the stems. Very early signs of larval
feeding indicate that other eggs will be hatching soon.
Use two insecticide applications 7 days apart to
control The squash vine borer is a key pest of winter
squash, gourds and pumpkins in Kentucky.
Unfortunately, it is hatching larvae and continue to
monitor for additional activity. Sprays need to
penetrate the canopy to cover the vines to be effective.
The squash vine borer is a key pest of winter squash,
gourds and pumpkins in Kentucky. Unfortunately, it
is usually noticed only after it has done its damage.
Symptoms appear in mid-summer when a long
runner or an entire plant wilts suddenly. Adult moths
begin to emerge about the time the plants begin to
run, and moth flight continues through mid August.
Only the unmated adult bugs overwinter in Kentucky.
The key to squash vine borer management is
controlling the borers before they enter the stem. Once
inside the vine, insecticidal control is ineffective. Poor
timing of sprays is the usual cause of inadequate
control. Monitor plants weekly from mid-June
through August for initial signs of the borer's frass at
entrance holes in the stems. Very early signs of larval
feeding indicate that other eggs will be hatching soon.
Use two insecticide applications 7 days apart to
control The squash vine borer is a key pest of winter
squash, gourds and pumpkins in Kentucky.
Unfortunately, it is hatching larvae and continue to
monitor for additional activity. Sprays need to
penetrate the canopy to cover the vines to be effective.



