
After this issue, the Kentucky Pest News will be
published on an alternate week basis. Weekly issues will
return in the spring.


 Diplodia ear rot is showing up in some corn fields at
rather high levels, with up to 25% of ears damaged. This
ear rot appears as a white, cottony fungal growth
between kernels, almost always progressing from the ear
shank upward and from the cob outward. Occasionally,
infections progress downward from the tip of the cob.
Severely affected ears can be obvious from a distance, as
the husks turn brown and dry down well before the rest
of the plant.
Diplodia ear rot is showing up in some corn fields at
rather high levels, with up to 25% of ears damaged. This
ear rot appears as a white, cottony fungal growth
between kernels, almost always progressing from the ear
shank upward and from the cob outward. Occasionally,
infections progress downward from the tip of the cob.
Severely affected ears can be obvious from a distance, as
the husks turn brown and dry down well before the rest
of the plant.
General Biology
The fungus that causes this disease, Stenocarpella maydis
(and in some cases, possibly Stenocarpella macrospora),
survives in previously disease ears and stalks of corn.
Thus, the fields with the highest risk are those in no-till
continuous corn; the lowest risk is in fields with 2-3
years away from corn with some tillage during the
rotation. Sometimes outbreaks can be associated with
unusually susceptible cultivars, though all cultivars can
be infected under high enough disease pressure.
Although the fungi that cause Diplodia ear rot are now
considered to be members of the genus Stenocarpella,
they were once considered to be species of Diplodia. For
simplicity's sake, the name of the disease itself, Diplodia
ear rot, has been retained.
The good news about Diplodia ear rot, if there is any, is that this particular ear rot is not known to be associated with mycotoxins (toxins produced by fungi). So while the disease does damage the grain, sometimes severely, which results in a loss of feed quality, there are no toxin concerns with this grain in the U.S.
Where severe outbreaks are observed, we would advise sending a sample to the UK Plant Diagnostic Laboratory in Lexington or Princeton, so that the species of Stenocarpella can be identified. This information could be useful to advance breeding for resistance.
Discounts Possible
Diplodia ear rot can potentially result in significant
discounts when graded at the elevator or other point of
sale. This is due to several factors.
(1) Diseased kernels are lighter, which can reduce test weight and thus result in a test-weight discount. While test-weight discounts vary by elevator, they are commonly around 1 cent/bu for every pound below 54 lb/bu.
(2) Diplodia-affected kernels will also be counted among the Total Damaged Kernels tally. Damage discounts vary by elevator but are usually around 1 cent/bu for every percent of total damage above 5%. Loads with over 20% damage may be subject to rejection.
(3) Diplodia infection also results in more cobs and kernels being ground up during shelling, resulting in a higher level of broken corn and foreign matter (BCFM). While BCFM discounts vary by elevator, they are commonly around 2 cent/bu for every percent of BCFM above 3%. Loads with foreign matter over 10% may be subject to rejection.
Storage Considerations
Infected kernels break easily during shelling and
handling, which can result in more fine material in the
bin. To circumvent this we recommend cleaning to
remove fines, especially after drying and before delivery
and/or storage.
Although drying to 15% will stop growth of Stenocarpella in kernels, the rot has opened the kernel up to invasion by other storage fungi that can grow at 15% moisture, such as Aspergillus glaucus. Thus, corn affected by Diplodia ear rot should be dried to below 14%, cleaned, and cooled to below 50 F as soon after harvest as possible. Also, do not store Diplodia-affected corn into the summer.
Producers should test the nutritional value of the affected corn before feeding to livestock. Mycotoxins are not a concern but that would help producers adjust the rations to compensate for any quality loss from the ear rot.
More information on the biology and management of Diplodia ear rot can be found at the University of Kentucky Extension Plant Pathology publications homepage at http://www.ca.uky.edu/agcollege/plantpathology/PPAExten/pppublin.htm.
 Leaf diseases such as gray leaf spot and northern leaf
blight are reportedly continuing to progress in many
corn crops. This has already started to result in
problems with stalk strength, as the corn plant fills the
grain by drawing reserves from the stalk. Producers are
advised to scout all fields with significant levels of leaf
disease for stalk rots by either pinching the lower 2 or 3
stalk internodes, or by pushing stalks 10 to 12 inches
from vertical to check for lodging. Harvest early if 10 to
15% show weak stalks, before a strong wind leaves the
crop on the ground.
Leaf diseases such as gray leaf spot and northern leaf
blight are reportedly continuing to progress in many
corn crops. This has already started to result in
problems with stalk strength, as the corn plant fills the
grain by drawing reserves from the stalk. Producers are
advised to scout all fields with significant levels of leaf
disease for stalk rots by either pinching the lower 2 or 3
stalk internodes, or by pushing stalks 10 to 12 inches
from vertical to check for lodging. Harvest early if 10 to
15% show weak stalks, before a strong wind leaves the
crop on the ground.
For information about corn pests, visit
"Insect Management Recommendations".



In the August 2, 2004 issue of the Kentucky Pest News
http://www.uky.edu/Agriculture/kpn/kpn_04/kpndx_04.htm
, I presented an article written by Cindy
Finneseth, Seed Testing Coordinator, UK Seed Testing
Laboratory, indicating that the 2004 Fusarium Head
Blight (FHB) epidemic had seriously impacted
germination of many seed lots. Also, the article indicated
that lots commonly have high levels of infection by
Fusarium graminearum, the cause of FHB. The article
makes it clear that treating seed with certain fungicides,
such as Raxil-Thiram, among others, can bring
moderately substandard germination seed lots up to
acceptable levels (84% and higher). Still, based on some
questions I am receiving, there is concern by some
producers that planting seed infected by F. graminearum
this fall may increase the risk that FHB will be more
severe next spring.
The short answer to the above concern is that there is no
relationship between F. graminearum levels in seed and
the amount of FHB that will occur the following spring.
F. graminearum in seed is exclusively a
germination/stand consideration. This is because spores
of F. graminearum that infect wheat heads in the spring
and cause FHB, originate from infested crop residue,
primarily corn, and not seed or seedlings of the current
wheat crop. Thus, if weather conditions are highly
conducive to spore production and infection next spring,
serious FHB is likely to occur even in fields planted to
Fusarium-free seed the previous fall. Conversely, if the
weather does not favor either situation, low FHB levels
are likely even if seed planted had high levels of
infection. The bottom line is that there is no reason to
fear planting seed with high levels of F. graminearum as
long as germination, either before or after treating seed
with an appropriate fungicide, is at an acceptable level.
See "Insect Management Recommendations" for more wheat pest information.


Peach growers in some regions of Kentucky are observing increased levels of a fruit rot that appears to be new to our growers. The disease, called anthracnose, or sometimes referred to as crater rot, is caused by a fungus called Colletotrichum gloeosporioides. It was first noticed on a few peach trees in a western Kentucky orchard a few years ago and is now appearing in other orchards as well. The fruit rot disease appears to affect nearly all peach cultivars and may appear continuously from June through August.
--Symptoms. The peach anthracnose being observed here is characterized by circular, sunken, tan lesions on ripening fruit. The sunken lesions have a glistening or slimy surface, unlike brown rot disease with which it could be confused. Lesions may begin as small chlorotic spots on the fruit surface and gradually enlarge so that the rotted tan spot is visible. The decay continues to enlarge to an inch or two inches in diameter by which time the fruit is significantly decayed. Decay can be quite extensive in fruits on the tree and they continue to rot after harvest. When the decay becomes quite advanced, the fruit surface eventually takes on a grayish black color. Leaf and twig symptoms are generally not seen with this disease.
The fungus we have isolated from current season fruit decay appears to produce spores slightly smaller than those reported for C. gloeosporioides, but they are the same shape and appear to be unlike a related fungus, C. acutatum which also causes peach anthracnose. However, these fungi are sometimes difficult to tell apart. Similar Colletotrichum fungi also cause bitter rot of apple and anthracnose of strawberry.
--Disease development. The fungus overwinters in mummified fruit and in twig and bark cracks and crevices. The fungus Colletotrichum has a wide host range including herbaceous annuals and perennials as well as woody plants and other fruit crops. Because of this wide host range, the disease can become easily established in the orchard. Fungal spores are spread through splashing rain and infections are favored by warm, moist weather. Although symptoms are found primarily on ripe or ripening fruit, infections could occur during bloom and the fungus could then remain latent in developing fruit. The reason for outbreaks of the disease in Kentucky this year could be related to wet spring and summer weather.
--Disease management. Sanitation, the removal of
mummies and rotted fruit from the orchard, should be
helpful in reducing disease. Removal of wild Prunus
species growing near the orchard should also be helpful.
Anthracnose disease management with fungicides is
difficult because many of the widely used brown rot
fungicides such as sulfur are not very effective against
this disease. Captan, a fungicide registered for peaches,
has activity against anthracnose and should suppress the
disease. A combination of Elite (tebuconazole) and a
strobilurin-containing fungicide such as Pristine (both
registered for peaches) may also help and could be
alternated with captan. Fungicides need to be applied
season long from bloom to harvest.


You'll know a mole cricket when you see one. Not many creatures look more like a mole than this insect. From the shovel-like front legs to the brown, streamlined body, it is clearly built for efficient life in a tunnel. Mole crickets usually live in light sandy, or loamy soils that chronically have a high moisture content.
Mole crickets burrow through the soil in search of food, mostly small insects. Their tunneling can cause minor damage but rarely justifies control. Above average rainfall means a moister soil profile and conditions conducive to mole cricket activity.
 Large, furry, red and black velvet ants are crawling
across some lawns now. While resembling ants, these
insects belong to a family of solitary wasps. Female
velvet ants lay their eggs in the nests of bees and wasps.
Their larvae feed on the bee and wasp grubs developing
in the nest.
Large, furry, red and black velvet ants are crawling
across some lawns now. While resembling ants, these
insects belong to a family of solitary wasps. Female
velvet ants lay their eggs in the nests of bees and wasps.
Their larvae feed on the bee and wasp grubs developing
in the nest.
Velvet ants pack a painful sting which they will use to defend themselves if they are bothered but they are not aggressive or "mean". There is no central nest, individual ants live on their own; thus, there is no place to focus control.
For more information, see
http://www.uky.edu/Agriculture/Entomology/entfacts/trees/ef442.htm


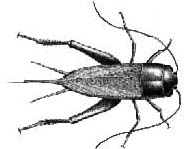 "Hundreds of black, � -inch long bugs are hopping out of my
grass, flower beds, and onto my patio . When I open the
garage door in the morning, a bunch more jump inside. What
are these critters and how do I get rid of them? Several
homeowners have called with this complaint in recent
weeks. The culprits are field crickets.
"Hundreds of black, � -inch long bugs are hopping out of my
grass, flower beds, and onto my patio . When I open the
garage door in the morning, a bunch more jump inside. What
are these critters and how do I get rid of them? Several
homeowners have called with this complaint in recent
weeks. The culprits are field crickets.
Warm, moist conditions often produce outbreaks of field crickets during late summer in Kentucky. Infestations are especially common in thatchy lawns and around buildings that are heavily mulched, landscaped or overgrown. Crickets lay their eggs in moist soil. The immatures (nymphs) pass through several stages (instars), and there may be 1 to 3 generations per year.
Management -- Field crickets are primarily a nuisance; they do not bite, transmit diseases or infest foodstuffs. Since they are dependent upon moisture, they typically do not survive indoors more than a few days. One option is to do nothing other than vacuum or sweep up those that manage to get inside. Removing excess mulch (a 2 to 3-inch layer is plenty for landscaping), weeds and debris close to the foundation will make the area less attractive to crickets. Installing tight-fitting door sweeps, sealing cracks, and performing other forms of exclusion (see Entfact-641, How to Pest-Proof Your Home) will further limit the entry of crickets, spiders, ground beetles and other unwanted pests.
For clients demanding immediate relief, pest proofing can be supplemented with exterior insecticide treatment. Homeowners will get the most for their efforts by applying longer-lasting pyrethroid products (e.g., Bayer Advanced� Lawn & Garden Multi-Insect Killer Concentrate, Spectracide Triazicide Soil & Turf Insect Killer Concentrate). Sevin (carbaryl) also is effective. Apply with a pump up or hose end sprayer, treating along the bottom of exterior doors, lower edge of siding, and around the outside perimeter of the foundation in a 2 to 10-foot wide band along the ground, and 2-3 feet up the foundation wall. Pay particular attention to the crack where grass meets the foundation.
Homeowners or businesses who choose not to tackle
these activities may wish to hire a professional pest
control firm. Field cricket problems subside with the
onset of cooler weather.


 Grubs damage muscle tissue along the back line of cattle
and cut holes in the hide to breathe. The results of their
handiwork aren't visible until February or March but
now is the time for applying the preventive treatment
that will keep them under control.
Grubs damage muscle tissue along the back line of cattle
and cut holes in the hide to breathe. The results of their
handiwork aren't visible until February or March but
now is the time for applying the preventive treatment
that will keep them under control.
Kentucky cattle should be treated for grubs between July 31 and October 15. During this period the grubs are very small and vulnerable. Animal complications can occur if treatments are applied later, when the grubs are in esophagus or spinal cord tissue.
Use care when treating for cattle grubs. Accurate animal weights allow economical and effective treatment. Over- dosing wastes money and may cause animal stress while under-dosing may result in unsatisfactory control.
For more information livestock pests, visit "Insect Management Recommendations".


Recent samples in the Diagnostic Lab have included gray leaf spot on field corn; sudden death syndrome, Fusarium root rot, downy mildew, frogeye leaf spot, potassium deficiency and low pH problems on soybean; black shank, blue mold, frogeye leaf spot, target spot, hollow stalk, Fusarium wilt, Fusarium stem canker, frenching, tobacco streak virus, and weather fleck on tobacco.
On fruits and vegetables, we have diagnosed double blossom on blackberry; anthracnose on bean; bacterial spot on pepper; Microdochium blight on squash; and Septoria leaf spot, buckeye rot, blossom end rot, stinkbug injury and early blight on tomato.
On ornamentals and turf, we have seen Pythium root rot
on geranium; Alternaria leaf spot on impatiens;
Rhizoctonia root rot on penta and zinnia; black root rot
on petunia; Phytophthora aerial blight, Pythium root rot,
and black root rot on vinca; powdery mildew and
transplant shock on dogwood; Verticillium wilt on
blackgum and maple; bacterial scorch and Actinopelte
leaf spot on oak; Cercospora leaf spot on honeylocust;
Rhizosphaera needle cast on spruce; Phomopsis canker
on willow; and gray leaf spot, Helminthosporium leaf
spot, and loose smut on turf.


UKREC-Princeton, KY, August 6-13 , 2004| Black Cutworm
| 1
| True Armyworm
| 4
| Corn Earworm
| 22
| European Corn Borer
| 6
| Southwestern Corn Borer
| 1
| Fall Armyworm
| 4
| | |
To view previous trap counts for Fulton County, Kentucky go to - http://ces.ca.uky.edu/fulton/anr/ and click on "Insect Trap Counts".
For information on trap counts in southern Illinois visit the Hines Report at - http://www.ipm.uiuc.edu/pubs/hines_report/index.html. The Hines Report is posted weekly by Ron Hines, Senior Research Specialist, at the University of Illinois Dixon Springs Agricultural Center
NOTE: Trade names are used to simplify the information presented in this newsletter. No endorsement by the Cooperative Extension Service is intended, nor is criticism implied of similar products that are not named.
Lee Townsend
Extension Entomologist