Kentucky Pest News Newsletter
HIGHLIGHTS IN THIS ISSUE
Number 898 __________ Sep 25, 2000
TOBACCO
CORN
WHEAT
SHADE TREES AND ORNAMENTALS
HOUSEHOLD
PESTICIDE NEWS AND VIEWS
DIAGNOSTIC LAB HIGHLIGHTS
IPM TRAP COUNTS
ANNOUNCEMENTS
TOBACCO
DEALING WITH MOLD ON CURING TOBACCO
By William Nesmith*, George Duncan and Gary Palmer
 Recently, some areas of Kentucky have experienced
periods of hot, humid weather with heavy fog
persisting from dark to mid-morning for several days
running. Others have experienced several days of
misty to heavy rain. Now reports are being received of
extensive molding of cured tobacco, especially in river
valleys. The problem is generally not occurring on
uncured tobacco. Rather, it is mainly associated with
tobacco harvested early and already cured, which in
general was some of the best quality leaf from this
year's crop.
Recently, some areas of Kentucky have experienced
periods of hot, humid weather with heavy fog
persisting from dark to mid-morning for several days
running. Others have experienced several days of
misty to heavy rain. Now reports are being received of
extensive molding of cured tobacco, especially in river
valleys. The problem is generally not occurring on
uncured tobacco. Rather, it is mainly associated with
tobacco harvested early and already cured, which in
general was some of the best quality leaf from this
year's crop.
The frequent periods of protracted mild, wet weather
during August and September have been providing
favorable conditions for mold development in curing
tobacco, especially that housed early. With more wet
weather in the forecast, growers should expect
increased potential for mold development in mid to
late harvests, as more of the tobacco cures and
becomes more susceptible to mold development. Let's
all remember that a greater percentage of the 2000
crop is earlier than normal, so there is a higher
percentage of this crop exposed to the mold threat.
Thus far, County Extension Agents have indicated
that most mold activity has been associated with
tobacco housed in traditional barns, but tobacco
housed early in out-door curing systems is also
developing mold. However, this pattern may be as
much the result of the tobacco being further along in
the curing process, because in general, growers filled
the barns first. On some farms where out-door
structures were used in early August, significant mold
has been reported, especially where the structures are
located in low areas. For example, Mike Carter,
Garrard County Extension Agent, reported
considerable house burn and mold developing in
outdoor structures in his area.
Some frequent questions are: Why does mold develop
some years and not others? How do you prevent mold
development? Does mold harm the tobacco? And,
how do you remove it from cured leaf? Hopefully, the
discussion below will help growers and agents with
these questions.
Once the tobacco leaf is dead it becomes an excellent
source of food for a number of fungi that could not
feed on it while it was alive. Thus, the pattern of the
mold being mainly associated with early housed
tobacco relates to the fact that it has more dead leaf,
while the later-housed plants are still alive. The
colonization of the leaf by these fungi results in moldy
tobacco which is lower in quality and weight due to
deterioration, off-colors, and residues from fungal
metabolism. Consequently, moldy tobacco is of less
value.
A small amount of mold develops every year on cured
tobacco, especially the stems (mid-ribs), but this is not
usually considered to be serious. However, when
mold develops on the lamina of the leaf, there is
concern. Problem levels of mold develop when moist
conditions prevail. Furthermore, the higher the
temperature during the moist period, the more rapid
is the mold development. In general, the mold growth
rate doubles for every 18 degrees F in increased
temperature.
Most of the mold problems thus far are associated
with early crops in poorly managed curing situations,
such as tobacco hanging close to the ground or in
areas where surface water is draining into the barn. If
the humid weather being experienced continues to
persist, the mold problem could begin to plague
tobacco housed under better curing conditions and
even crops housed later in the season.
Judicious management of the barn's ventilation
system is critical to preventing moldy tobacco.
Basically, it boils down to controlling moisture in the
dead leaf. The leaf absorbs water from the humid air
around it, so the key is to minimize the time moisture-
laden air exists in the barn, especially when
summer-like temperatures are occurring.
Generally, the moisture content of air is higher at
night and during rainy periods than during the
daytime. Therefore, closing the barn doors and
ventilators at night and promptly before a rainy
period should reduce the level of moisture inside the
barn. Opening the ventilation system after the rain
stops and during daylight hours will allow the inside
air to dry more rapidly. Obviously, proper barn
construction is critical to this operation, including
good site selection and adequate ventilation. Fans can
be used to increase ventilation capabilities, (See
AEU-7, `Using Fans in Conventional Burley Barns').
However, it is important that enough air be moved
through the tobacco and not just in the gable ends or
at the ground level. With outdoor or field curing
structures, plastic covers need to be in place to prevent
direct wetting of the leaf, followed by proper
manipulation of plastic to manage ventilation,
followed by timely stripping of the crop from these
structures.
When wet periods persist longer than 24-36 hours,
especially when temperatures are warm, the inside air
can become saturated even with ventilators closed.
Supplemental heat, although costly and probably not
economical at current prices, can be used to dry the
air. If heat is being used, the ventilation system should
be partially opened to allow the warm moist air to
escape the barn. Otherwise, the barn and tobacco will
`sweat'. Once the favorable outside drying conditions
return the heat should be stopped and the barn
opened. If heat is not used during these prolonged
wet weather periods, the next best option is to leave
the barn open to take advantage of whatever air
circulation and drying exist.
Removing Mold from Tobacco
No chemical is labeled for preventing mold growth on
tobacco in the curing barn. But there also is no
shortage of rumors of home remedies being tried.
Most of those could result in serious and illegal
residue problems that would destroy the usefulness of
the tobacco to the industry. Spraying Ridomil on the
barn floor, walls, and hanging tobacco does NOT
prevent molding of tobacco based on properly
conducted experimentation; besides, it is not labeled
for such use. This fungicide does not even control the
group of fungi involved!
There is a badly out-of-date publication from back in
the 1960 or 70's that many have. It clearly indicates
that experiments were conducted several years ago in
cooperation with the University of Kentucky which
demonstrated that mold could be removed to some
extent from cured tobacco with a variety of alcohols.
The alcohol compounds mentioned in that publication
are exempt by EPA from residue tolerance
requirements on tobacco, so they can be used in
conjunction with otherwise good agricultural
practices. HOWEVER, PLEASE BE AWARE THAT no
company has come forward and labeled such use, so
the liability associated with their use rests with those
making the recommendation and those using them!
Alcohols are highly flammable and thus pose a
serious risk to persons and property from fire.
Therefore, we specialists are not willing to
recommend the use of alcohols for the removal of
mold on cured tobacco. We mention the alcohols
here, not as some type of hidden recommendation,
but as point of clarification and because many have
requested our position on this issue.
* William Nesmith, George Duncan and Gary Palmer
are Extension Plant Pathologist, Extension
Agricultural Engineer, and Extension Tobacco
Specialist, respectively, in the College of Agriculture,
University of Kentucky.

CORN
DIPLODIA EAR ROT UPDATE
By Paul Vincelli, Plant Pathologist and Sam McNeill, Ag Engineer
 Diplodia ear rot has been a significant problem in
some corn fields in Kentucky this year. Thanks to
Extension Ag Engineer Sam McNeill, I became aware
of a new publication on Diplodia ear rot from Purdue
University(
Diplodia Ear Rots in Indiana, Grain
Quality Fact Sheet #45
, by C. Woloshuk and D.Maier). Their publication has some important points
which are summarized here.
Diplodia ear rot has been a significant problem in
some corn fields in Kentucky this year. Thanks to
Extension Ag Engineer Sam McNeill, I became aware
of a new publication on Diplodia ear rot from Purdue
University(
Diplodia Ear Rots in Indiana, Grain
Quality Fact Sheet #45
, by C. Woloshuk and D.Maier). Their publication has some important points
which are summarized here.
Discounts Possible
Diplodia ear rot can potentially result in significant
discounts when graded at the elevator or other point
of sale. This is due to several factors.
(1) Diseased kernels are lighter, which can reduce test
weight and thus result in a test-weight discount.
While test-weight discounts vary by elevator, they are
commonly around 1 cent/bu for every lb/bu of test
weight below 54 lb/bu.
(2) Diplodia-infected kernels will also be counted
among the Total Damaged Kernels tally. Damage
discounts vary by elevator but are usually around 1
cent/bu for every percent of total damage above 5%.
Loads with over 20% damage may be subject to
rejection.
(3) Diplodia infection also results in more cobs and
kernels being ground up during shelling, resulting in
a higher level of broken corn and foreign matter
(BCFM). While BCFM discounts vary by elevator,
they are commonly around 2 cent/bu for every
percent of BCFM above 3%. Loads with foreign
matter over 10% may be subject to rejection.
Storage Considerations
Infected kernels break easily during shelling and
handling, which can result in more fine material in the
bin. To circumvent this we recommend cleaning to
remove fines, especially after drying and before
delivery and/or storage.
Although drying to 15% will stop Diplodia growth in
kernels, the rot has opened the kernel up to invasion
by other storage fungi that can grow at 15% moisture,
such as Aspergillus glaucus. Thus, corn affected by
Diplodia should be dried to below 14%, cleaned, and
cooled to below 50 F as soon after harvest as possible.
Also, do not store Diplodia-affected corn into the
summer.
WIDESPREAD STALK ROT PROBLEMS MEANS PRODUCERS SHOULD SCOUT NOW
By Paul Vincelli
In the last issue, I reported on cases of severe stalk rot
in the Green River Area. Since that time, reports of
moderate to severe stalk lodging have come in from a
number of locations. Because of the widespread
occurrence of stalk lodging problems, producers are
strongly encouraged to scout all fields for lodging
potential. A simple way to do this is to walk through
the field and push stalks 8-10 inches from vertical:
those that fail to spring back have the potential to
lodge in a strong wind. If more than 10-15% of the
stalks in a field lodge by this method, producers may
wish to consider scheduling the field for early harvest.
Even though the cost of liquid propane is high,
increasing the cost of drying early-harvested fields,
the risk of lodging and the resulting problems and
yield loss must also be considered.
We have examined some cases of stalk lodging in the
Diagnostic Labs. At this point it appears as though
Gibberella stalk rot is the most common disease
involved. Diplodia stalk rot has been found in several
instances, and Fusarium stalk rot has also been
confirmed in at least one instance. While injury from
stalk-boring insects is often present in many fields, for
the cases in which I've been involved, most of the
stalks have lodged as a result of stalk rots, not insect
injury. This is important for producers to understand
so that they don't overuse Bt corn hybrids next year in
an effort to control the wrong problem. UK
recommendations for use of Bt hybrids are still the
best guidelines.
Stalk rot diseases are the result of opportunistic
infections. What this means is that certain stress
factors, such as low carbohydrate status in the stalk,
predispose the plant to infection, and the "first one to
attack, wins"; that is, the first fungus to infect the stalk
is the one that causes the stalk weakening. What this
also means is that the most important thing to do from
a production standpoint when stalk rots attack is to
evaluate one's cultural program and see if there are
particular agronomic stresses that might be alleviated.
Factors that might enhance stalk rot problems include:
excessive plant population, excessive N in relation to
potash, high N levels early in the season followed by
N loss through leaching or denitrification, inadequate
levels of potash, low stalk strength ratings of hybrids
planted, and severe leaf disease. Producers may wish
to evaluate these factors to see if there are ways to
reduce the risk of stalk lodging in future years.
CORN BORER ADVISORY
By Ric Bessin
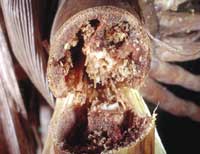 Reports of heavy corn borer infestations continue,
some corn producers say this is the worst they have
seen in 20 years. Much of this is caused by
southwestern corn borer, but there is considerable
damage by European corn borer as well.
Southwestern corn borer is continuing to girdle stalks
just above the soil line and will continue to girdle additional
stalks through mid October. Producers need to
identify the most heavily infested fields and plan to
harvest them as soon as the moisture level of the grain
is acceptable. Pay particular attention to fields planted
after May 10.
Reports of heavy corn borer infestations continue,
some corn producers say this is the worst they have
seen in 20 years. Much of this is caused by
southwestern corn borer, but there is considerable
damage by European corn borer as well.
Southwestern corn borer is continuing to girdle stalks
just above the soil line and will continue to girdle additional
stalks through mid October. Producers need to
identify the most heavily infested fields and plan to
harvest them as soon as the moisture level of the grain
is acceptable. Pay particular attention to fields planted
after May 10.
For more information about corn pests, visit
"Insect Management Recommendations".

WHEAT
POWDERY MILDEW MANAGEMENT: NOW IS THE TIME FOR DECISION-MAKING
By Don Hershman
 Powdery mildew, if left to develop unchecked, can
seriously impact crop yield. This is especially true for
fields where disease develops early (e.g., pre-flag leaf
emergence) and continues throughout the season.
When significant levels of powdery mildew are
detected for a field, impacted growers may consider
the option of disease management using either Tilt or
Quadris. Either fungicide will do the job, but if
applications are made before the early heading stages,
late-season disease control may be compromised. The
only option would be to make a second application
using Quadris. It is illegal to apply more that 4 fl oz/A
of Tilt to a crop, but the Quadris label does provide for
two applications. Excessive cost would, however,
assures that this option is not a viable one.
Powdery mildew, if left to develop unchecked, can
seriously impact crop yield. This is especially true for
fields where disease develops early (e.g., pre-flag leaf
emergence) and continues throughout the season.
When significant levels of powdery mildew are
detected for a field, impacted growers may consider
the option of disease management using either Tilt or
Quadris. Either fungicide will do the job, but if
applications are made before the early heading stages,
late-season disease control may be compromised. The
only option would be to make a second application
using Quadris. It is illegal to apply more that 4 fl oz/A
of Tilt to a crop, but the Quadris label does provide for
two applications. Excessive cost would, however,
assures that this option is not a viable one.
In the old days, powdery mildew could be effectively
and decisively controlled by applying 2 oz of
Bayleton. You may recall that Bayleton no longer has
a wheat label. Thus, that option no longer exists with
one exception. I am told that it still may be possible to
find old Bayleton with wheat on the label. I have no
idea where it is or how easy or hard it would be to
come up with any significant stocks of Bayleton. But if
it can be found, it is legal to use on wheat even though
new Bayleton cannot be applied.
There are two ways to avoid the powdery mildew
fungicide dilemma altogether. The most economical
and, perhaps, desirable way is to plant a variety with
resistance to powdery mildew. These varieties are
readily available and perform very well in the
presence of significant disease pressure. Another way,
but one which is significantly more costly and
involved logistically, is to plant seed treated with the
fungicide Baytan. Baytan seed treatment will
effectively control powdery mildew in a crop up to the
boot stage of development in the spring. This is
usually all it takes to stifle yield reductions due to
powdery mildew in a disease-susceptible variety. The
cost of Baytan and arranging to have seed treated with
the product makes the use of a resistant variety more
palatable.
With the wheat planting season just around the
corner, it would be best to address the powdery
mildew situation now while you still have all the
options discussed above available to you.

SHADE TREES AND ORNAMENTALS
A KENTUCKY CONFERENCE ON TREE ROOTS AND SOIL HEALTH
By John Hartman
"Down and Dirty in Kentucky: a closer look at tree
roots and soil health," is the theme of this year's
Kentucky Urban and Community Forestry
Conference. The Conference, sponsored by the
Kentucky Arborists Association, the Kentucky
Division of Forestry, and the U.K. Cooperative
Extension Service is scheduled for Mon., October 30 -
Wed., November 1 at the Holiday Inn, (405 Wilkinson
Blvd.) Frankfort. The program should prove to be
very informative with several renowned tree and soil
experts from across the country listed as prominent
speakers.
On Monday, topics such as "Making Tree Roots
Visible," "Soil Food Web," "Soil Structure," "Dr.
Treevorkian," and "Pesticide Applicator Training"
will be featured. Speakers include Bonnie Appleton,
Serita Frey, Chris Carlson, Jim Chatfield, and Monte
Johnson. Tuesday, the subjects of "Soil-oriented
Construction Damage," "Planting Depth Study,"
"Tree Care Advisor Program," "Root Diseases,"
"Street Trees," and "Repairing Soils After
Development" will be covered by presenters such as
Alan Siewert, Bill Fountain, John Hartman, Marty
Shaw, and Gary Johnson. The Conference will
conclude Wednesday morning with "Soil Biology,"
"Soil Remediation," "Urban Soil vs. Forest Soil," and
"ISA Arborists Certification Exam" with presentations
by Kim Coder, Tom Kimmerer, and Dave Leonard.
In my experience, trees in landscapes suffer more
from soil and root-related problems than they do from
above-ground problems. Further, most soil-related
tree problems are the result of mis-management of
cultural practices such as soil preparation, planting
depth, root injuries, site selection, watering, mulch
application and species choices. County Extension
Agents, arborists, landscape maintenance
professionals, and municipal tree board members face
these and many other tree difficulties on a daily basis.
This conference will provide solutions to many of
these problems affecting tree health.
To obtain registration materials, contact Dino Kent,
Conference Chair, 215 Forest Park Road, Lexington,
KY 40503; tel. 859-278-3582; e-mail, treesec@msn.com.
For conference program information, contact Dave
Leonard, 1302 North Limestone, Lexington, KY 40505;
tel. 859-252-2529; e-mail dave7oaks@aol.com. For
trade show information contact Larry Lose, 10105
Afton Road, Louisville, KY 40223; tel. 502-245-4036, e-
mail lmlose@christianliving.net. Reservations for
lodging may be made at the Holiday Inn, 405
Wilkinson Blve., Frankfort, KY 40601; tel. 502-227-
5100.
BACTERIAL LEAF SCORCH IS VISIBLE NOW
By John Hartman
County Extension Agents, landscape professionals,
and observant homeowners have no doubt noticed
premature browning of many shade trees, particularly
oaks this month. Most of the affected trees are
suffering from bacterial leaf scorch, caused by Xylella
fastidiosa. This disease is found on several shade and
landscape trees in Kentucky, but oaks, especially pin
oaks are the primary victims.
Symptoms. Bacterial leaf scorch disease symptoms
include premature leaf browning and defoliation, and
leaves have a marginal necrosis. Symptoms appear in
the summer and early fall and may begin on one
branch. Infected trees leaf out normally the following
year, and in late summer, leaves of a few more
branches turn prematurely brown. These events
repeat themselves over a period of several years until
the entire infected tree turns prematurely brown in fall
and begins to show twig and branch dieback and tree
decline. The reason for the scorching symptom is that
the pathogen is found in the xylem tissues of trees
where it blocks the flow of water to the leaves. The
bacterial leaf scorch pathogen is spread by
leafhoppers and treehopper insects, but it does not
appear to be spread from tree to tree very rapidly by
these insects. Nevertheless, in some neighborhoods
with mature oaks and with the disease present for
many years, perhaps half of the trees can show
symptoms of bacterial leaf scorch. The disease is
confirmed in the University of Kentucky Plant Disease
Diagnostic Laboratory by detecting the bacteria in
infected leaves with a special test specific for Xylella,
the cause of the disease.
Where is bacterial leaf scorch? In Kentucky, bacterial
leaf scorch has been commonly observed in pin oak,
red oak, shingle oak, bur oak, and sycamore. It is also
occasionally found in red maple, sugar maple, silver
maple, London plane, and sweetgum. In other states,
the disease has also been detected in American elm
and mulberry. On the U.S. capitol mall in
Washington, DC, for example, as early as mid-June,
American elms can be seen with symptoms of
bacterial leaf scorch. Bacterial leaf scorch is present in
landscape trees in many of the urban areas of
Kentucky. The bacteria have been identified in urban
trees from such cities as Paducah, Madisonville,
Owensboro, Bowling Green, Somerset, Louisville, and
Lexington. In Kentucky, this disease has not been
detected in forest trees.
Prognosis for infected trees. There is no effective
long-term cure for bacterial leaf scorch. Because
infected trees decline gradually, it may take from five
to ten years until they have many dead limbs and
branches and need to be removed. In the meantime,
tree owners should provide good growing conditions
for the trees to prolong their survival, and begin to
plant replacement trees that will attain a reasonable
size before the diseased ones are removed.
BOXELDER BUGS
By Lee Townsend
 Boxelder bugs are common insects that feed on sap
from leaves, twigs, and seeds of boxelders, as well
other members of the maple family. Large numbers of
them can be seen on tree trunks, or branches, or
sunning themselves on the south or west sides of
buildings in the spring and fall. These harmless
accidental invaders may be a temporary nuisance as
they move into and out of sheltered overwintering
sites in the fall and spring.
Boxelder bugs are common insects that feed on sap
from leaves, twigs, and seeds of boxelders, as well
other members of the maple family. Large numbers of
them can be seen on tree trunks, or branches, or
sunning themselves on the south or west sides of
buildings in the spring and fall. These harmless
accidental invaders may be a temporary nuisance as
they move into and out of sheltered overwintering
sites in the fall and spring.
Adult boxelder bugs are elongate, 1/2" long insects with
sucking mouthparts. They are mostly black with
some red markings. There are three narrow red lines
on the segment behind the head, one down the center
and one on either side and a thin red inverted "V"
about the middle of the back. The wingless immature
or nymphal stage has a black head, antennae, and
legs. The red abdomen has an orange-yellow stripe
and spot down the center of the back.
Boxelder bugs feed on plants during the summer.
They move to sheltered areas (including nearby
houses and buildings) in the fall and remain there
during the winter. Sweep or vacuum up and discard
those found inside. They will not multiply nor will
they infest any household articles. These insects are
only a nuisance or accidental invader; however, they
may leave an odor and stain when crushed.
Exclusion is important in reducing numbers that get
indoors. Check door sweeps and places outside the
house that may allow them to get inside. If large
numbers are present every year, removal of female
boxelder plants should be considered because this is
the major host plant for them. Other alternatives
include controlling insects while they are on the
boxelder plant or direct sprays of congregations of the
insects on outside walls. Insecticidal soap provides a
safe control alternative but it must be sprayed directly
on the insects to be effective.

HOUSEHOLD
HOW TO PEST-PROOF YOUR HOME
By Mike Potter
 According to a statewide poll of Kentucky
householders, 93% expressed concern over finding
insects inside their home. More than half indicated
that a single cockroach, cricket, or spider would
prompt them to use a can of bug spray or call an
exterminator. What many people do not realize,
however, is that most pests discovered indoors have
flown or crawled in from outdoors.
According to a statewide poll of Kentucky
householders, 93% expressed concern over finding
insects inside their home. More than half indicated
that a single cockroach, cricket, or spider would
prompt them to use a can of bug spray or call an
exterminator. What many people do not realize,
however, is that most pests discovered indoors have
flown or crawled in from outdoors.
One of the best ways to limit unwanted intrusions by
insects, rodents, birds, squirrels and other pests is to
deny them entry a procedure known as pest
proofing. Many pests seek refuge in homes and
buildings in response to changes in weather, such as
extended periods of rain or drought, or the onset of
cooler autumn temperatures. Taking steps to block
their entry before they end up inside can greatly reduce
the chances of future sightings.
Outlined below are six useful tips for pest proofing
one's home or place of business. Steps 1-3 will also
conserve energy and increase the comfort level during
winter and summer. Equipment and materials
mentioned can be purchased at most hardware or
home improvement stores.
1. Install door sweeps or thresholds at the base of all
exterior entry doors. While laying on the floor, check
for light filtering under doors. Gaps of 1/16" or less
will permit entry of insects and spiders; 1/4"-wide
gaps (the diameter of a pencil) are large enough for
entry of mice; "" gaps are adequate for rats. Pay
particular attention to the bottom corners as this is
often where rodents and insects enter.
Apply caulk (see #3 below) along bottom outside edge
and sides of door thresholds to exclude ants and other
small insects. Garage doors should be fitted with a
bottom seal constructed of rubber (vinyl
weatherstripping seals poorly in cold weather). Gaps
under sliding glass doors can be sealed by lining the
bottom track with " to 3/4 inch-wide foam
weatherstripping.
2. Seal utility openings where pipes and wires enter the
foundation and siding, such as around outdoor
faucets, receptacles, gas meters, clothes dryer vents,
and telephone/cable TV wires. These are common
entry points for rodents, ants, spiders, yellowjackets
and other pests. Holes can be plugged with caulk,
cement, urethane expandable foam, steel wool, copper
mesh, or other suitable sealant.
3. Caulk cracks around windows, doors, fascia boards, etc.
Use a good quality silicone or acrylic latex caulk.
Although somewhat less flexible than pure silicone,
latex-type caulks clean up easily with water and are
paintable. Caulks that dry clear are often easier to use
than pigmented caulks since they don't show
mistakes.
Buy a good caulking gun. Features to look for include
a back-off trigger to halt the flow of caulk when
desired, a built-in "slicer" for cutting the tip off of new
caulking tubes, and a nail for puncturing the seal
within. Hardware stores sell guns with these features
for less than $10.00. Prior to sealing, cracks should be
cleaned and any peeling caulk removed to aid
adhesion. For a professional look, smooth the bead of
caulk with a damp rag or a moistened finger after
application.
4. Repair gaps and tears in window and door screens.
Doing so will help reduce entry of flies, gnats,
mosquitoes and midges during summer, and cluster
flies, lady beetles, and other overwintering pests in
autumn. Certain insects, in particular leafhoppers and
hackberry psyllids, are small enough to fit through
standard mesh window screen. The only way to deny
entry of these tiny insects is to keep windows closed
during periods of adult emergence.
5. Install 1/4-inch wire mesh (hardware cloth) over attic,
roof, and crawl space vents in order to prevent entry of
birds, bats, squirrels, rodents, and other wildlife. Be
sure to wear gloves when cutting and installing
hardware cloth, as the wire edges are razor sharp.
Invest in a chimney cap to exclude birds, squirrels,
raccoons and other nuisance wildlife.
6. Consider applying an exterior (barrier) insecticide
treatment. While sealing is the more permanent way to
exclude pests originating from outdoors,
comprehensive pest-proofing is labor-intensive and
sometimes impractical. For clients requiring an
alternative, pest proofing can be supplemented by an
exterior treatment with an insecticide. Homeowners
will get the most for their efforts by applying longer-
lasting liquid formulations containing synthetic
pyrethroids (e.g., Bayer Advanced Home/Garden
Multi-Insect Killer, Spectracide Bug Stop , Ortho
Home Defense System ) or microencapsulated, slow-
release Dursban, sold at hardware/lawn and garden
shops.
Apply with a pump up sprayer, hose end sprayer, etc.,
treating at the base of all exterior doors, garage and
crawl space entrances, around foundation vents and
utility openings, and up underneath siding. It may
also be useful to treat around the outside perimeter of
the foundation in a 2 to 6-foot-wide band along the
ground, and 2-3 feet up the foundation wall.
Clients who choose not to tackle these activities may
wish to hire a professional pest control firm. Many
firms now offer pest proofing as an adjunct to other
services. When all else fails, a vacuum cleaner or
broom is often the best response to the occasional bug
that wanders in from outdoors.

PESTICIDE NEWS AND VIEWS
TACO BELL TACO SHELL CONTROVERSY
By Ric Bessin
Last week it was reported that a group had found
traces of StarLink corn in Taco Bell taco shells. This is
significant as StarLink corn possess the Bt gene that
produces the Cry9C protein. StarLink corn was
approved for commercial production in 1998, but the
grain produced from StarLink hybrids are restricted
for use only as animal feed. StarLink corn is not
permitted for human consumption. It is too early as to
speculate on the cause of this contamination and
additional tests by an independent lab is needed to
confirm this contamination.
While StarLink is not approved for human
consumption, the other Bt corns including YieldGard,
NatureGard, Knock Out, Bt-Xtra, and Attribute are
approved for human consumption. These other Bt
corn events use the Cry1A(b) and Cry1A(c) proteins.
The reason why StarLink has not received approval
for human consumption is that it is a novel Bt protein
and it is more resistant to degradation in the digestive
systems of mammals. For those reasons, the FDA is
concerned that it MAY be an allergen. Currently, it
has not been proven that the Cry9C protein is or is not
a human allergen. Until the human allergenicity issue
is resolved, StarLink corn will not be approved for
human consumption.
However, grain from StarLink corn is approved for
use as animal feed. Is there a problem when humans
eat animals that have been feed StarLink corn? No,
definitely not. The Bt proteins are either broken down
in the animal or pass through the digestive system
entirely. So despite the uncertainty of consuming
StarLink directly, there is no problem associated with
consumption of animals that have been raised on
StarLink grain.
If this StarLink contamination of the taco shells is
confirmed, then it will be necessary to determine how
this happened. There are several different scenarios
which may caused this type of contamination. This
may include but is not limited to:
--failure to properly clean old seed planting
equipment resulting in a mixture of StarLink with
other seed,
--pollen drift from a nearby StarLink field to a food
grade corn field resulting in cross pollination,
--misidentification of seed or grain on the farm or of
grain in marketing channels,
--inadequate cleaning of harvest machinery, loading
and transportation equipment, or storage facilities,
--careless mixing of grain, or
--failure to understand current use restrictions with
StarLink corn.
While the immediate hazard posed by StarLink corn
in the taco shells may be minimal, this does raise the
issue of how well can we maintain the identity of
these different types of corn with our current
marketing infrastructure? We will hear more about
this issue as additional information becomes available.

DIAGNOSTIC LAB HIGHLIGHTS
DIAGNOSTIC LAB - HIGHLIGHTS
By Julie Beale and Paul Bachi
Recent field crop samples submitted to the Diagnostic
Lab have included Gibberella stalk rot, Fusarium ear
rot (F. moniliforme), gray leaf spot, and the virus
complex on corn; Cercospora leaf spot and
Rhizoctonia stem canker on alfalfa; charcoal rot,
anthracnose, stem canker, sudden death symdrome,
soybean cyst nematode and root knot nematode on
soybean. Fruit and vegetables samples have included
anthracnose on raspberry; bacterial canker on cherry;
scab and brown rot on peach; Fusarium fruit rot on
cushaw; and Rhizoctonia stem canker on cabbage.
Ornamental samples have been numerous and have
included Pythium and Rhizoctonia root rots on
daylily; bacterial spot on Heuchera; Rhizoctonia stem
rot on impatiens; Cladosporium leaf blotch on peony;
impatiens necrotic spot virus and southern blight on
vinca; rust and summer patch on bluegrass;
anthracnose and gray leaf spot on ryegrass; ash
yellows on ash; Phytophthora root rot and black root
rot on boxwood; Cercosporella leaf spot on mulberry;
bacterial scorch on oak; Cercospora leaf spot on
sourwood; rose rosette on cultivated rose; and
bacterial leaf spot on viburnum.

IPM TRAP COUNTS:
By Patty Lucas, University of Kentucky Research Center
Insect Trap Counts
|
| UKREC-Princeton, KY, August 25 - Sep 8, 2000
|
| Fall Armyworm
| 8
|
| Corn Earworm
| 35
|
| European Corn Borer
| 7
|
| Southwestern Corn Borer
| 51
|
ANNOUNCEMENTS
DECEMBER KENTUCKIANA CROP PRODUCTION PROGRAM
By Lee Townsend
The annual Kentuckiana Crop Production Seminar
will be held December 12 - 13, 2000 at the Radisson
Hotel, Evansville, IN. A CCA continuing education
program, three hours of continuing education credit
in either cropping systems management or soil and
water management, starts at 8 am. The Crop
Production Seminar runs from 1 pm to 5:10 pm on
December 12 and from 8 am until noon on December
13. The program will be submitted to the Division of
Pesticides Ky Department of Agriculture for
continuing education credit for commercial pesticide
applicators (Categories 1 and 12).
CURRENT STATE LOCAL NEED (24-C) REGISTRATIONS FOR KENTUCKY
By Kentucky Division of Pesticides
Section 24 (c) of the Federal Insecticide Fungicide and
Rodenticide Act (FIFRA) permits the Kentucky
Department of Agriculture to register federally
registered pesticides for some uses that are not on the
existing label. 24-c or State Local Need Labels, are
valid only in the state of issue. The applicator must
possess a copy of the state label when the pesticide is
applied. In Kentucky, 24-c labels are valid for three
calendar years.
When there is an existing or expected local or minor
pest problem, the Kentucky Department of
Agriculture will be permitted to register one or more
pesticide products if:
_ there is no EPA-registered pesticide for the use in
question.
_ the EPA-registered pesticide is not available or
cannot be obtained in sufficient quantity.
_ there is a suitable EPA-registered pesticide which, if
used in accordance with the label, would not be safe
or effective under the local conditions.
States cannot register:
_
pesticides on food crops that do not have an
established tolerance on that crop.
_
pesticides containing active or inert ingredients not
contained in any EPA- registered products.
_
pesticide products or uses affected by suspension or
cancellation based on human health, environmental,
or efficacy considerations.
_
pesticide products and/or uses previously denied
registration by EPA.
Special local needs registrations may be sought by
commodity groups, university, industry or others.
The pesticide manufacturer or formulator must,
however, be willing to support the effort and to
prepare the documentation needed to justify the
request. The registration is not effective for more than
90 days if disapproved by the EPA Administrator
within that time.
Product Name - Use: Command 4EC - winter and summer squash
SLN Number: KY-930002
Expiration Date: 12/31/02
Product Name - Use: Dithane DF* - tobacco seedlings for transplant
SLN Number: KY-940002
Expiration Date: 12/31/02
Product Name - Use: Command 4EC - cabbage
SLN Number: KY-960003
Expiration Date: 12/31/02
Product Name - Use: Poast - tobacco
SLN Number: KY-970002
Expiration Date: 12/31/02
Product Name - Use: Dual Magnum* - transplanted bell peppers
SLN Number: KY-990001
Expiration Date:12/31/02
Product Name - Use: Dual Magnum* - cabbage
SLN Number: KY-990002
Expiration Date: 12/31/02
Product Name - Use: Acrobat MZ - control of metalaxyl/mefanoxam-
resistant strains of blue mold in tobacco
SLN Number: KY-990003
Expiration Date: 12/31/02
Product Name - Use: Terrazole 35 WP - control of pythium root rot in
tobacco transplant float beds
SLN Number: KY-000001
Expiration Date: 12/31/02
Product Name - Use: Tracer* - control of budworms and hornworms on
tobacco
SLN Number: KY-000002
Expiration Date: 12/31/02
* Still within EPA's 90-day period to revoke.European
and southwestern corn borer levels are high
COMMERCIAL PESTICIDE TRAINING MEETINGS
By Lee Townsend
Commercial pesticide training meetings are scheduled
for September 20, 2000 at the Fayette County
Extension office in Lexington and October 12, 2000 at
the UK Research and Education Center in Princeton.
Training for Categories 1 (Ag Plant), 4 (Seed
Treatment), 10 (Demonstration and Research, and 12
(Pesticide Dealer) begin at 8:30 am and end at 11:40
am (local time).
Training for Categories 2a (Forest Pest) and 3
(Ornamental and Turf) begin at 9:45 am and end at
1:15 pm. Applicators or dealers certified in categories
10 and 12 with primary interest in these categories
should attend this session.
Testing for commercial certification will be held at
1:30 pm.
Call Monte Johnson (859) 257?6693 for meeting
information. Contact Darlene Thorpe (859) 257?5955
for training packets. If you have questions concerning
certification credits that you have earned or laws and
regulations, call the Division of Pesticides (Ky
Department of Agriculture) (502) 564?7274.
Visit the Pesticide Applicator Training page for the
latest information on training and testing
opportunities.
http://www.uky.edu/Agriculture/PAT/welcome.htm
30th ANNUAL UK PEST CONTROL SHORT COURSE- October 3-5, 2000 in Lexington
By Lee Townsend
The 2000 University of Kentucky Pest Control Short
Course will energize you with information and ideas
to move forward with confidence.
This year's 30th Anniversity conference is packed with
useful topics for owners, managers, and technicians
alike - a hard-hitting business session on legal,
insurance and regulatory issues of termite baiting;
surviving negligence/bodily injury claims marketing
strategies for small companies; IPM in schools; new
insights on rodent trapping, emerging construction
challenges; practical termite research - even a half-day
workshop on managing 18 bothersome pests seldom
mentioned at other conferences around the country,
such as silverfish, psocids, snakes and ladybugs!
See the full program on the
Ky Pesticide Applicator Training Web Site
Call Darlene Thorpe (859) 257-5955 for more
information.
Lee Townsend
Extension Entomologist
BACK
TO KY PEST NEWS HOME
 Diplodia ear rot has been a significant problem in
some corn fields in Kentucky this year. Thanks to
Extension Ag Engineer Sam McNeill, I became aware
of a new publication on Diplodia ear rot from Purdue
University(
Diplodia Ear Rots in Indiana, Grain
Quality Fact Sheet #45
, by C. Woloshuk and D.Maier). Their publication has some important points
which are summarized here.
Diplodia ear rot has been a significant problem in
some corn fields in Kentucky this year. Thanks to
Extension Ag Engineer Sam McNeill, I became aware
of a new publication on Diplodia ear rot from Purdue
University(
Diplodia Ear Rots in Indiana, Grain
Quality Fact Sheet #45
, by C. Woloshuk and D.Maier). Their publication has some important points
which are summarized here.
 Recently, some areas of Kentucky have experienced
periods of hot, humid weather with heavy fog
persisting from dark to mid-morning for several days
running. Others have experienced several days of
misty to heavy rain. Now reports are being received of
extensive molding of cured tobacco, especially in river
valleys. The problem is generally not occurring on
uncured tobacco. Rather, it is mainly associated with
tobacco harvested early and already cured, which in
general was some of the best quality leaf from this
year's crop.
Recently, some areas of Kentucky have experienced
periods of hot, humid weather with heavy fog
persisting from dark to mid-morning for several days
running. Others have experienced several days of
misty to heavy rain. Now reports are being received of
extensive molding of cured tobacco, especially in river
valleys. The problem is generally not occurring on
uncured tobacco. Rather, it is mainly associated with
tobacco harvested early and already cured, which in
general was some of the best quality leaf from this
year's crop.

 Reports of heavy corn borer infestations continue,
some corn producers say this is the worst they have
seen in 20 years. Much of this is caused by
southwestern corn borer, but there is considerable
damage by European corn borer as well.
Southwestern corn borer is continuing to girdle stalks
just above the soil line and will continue to girdle additional
stalks through mid October. Producers need to
identify the most heavily infested fields and plan to
harvest them as soon as the moisture level of the grain
is acceptable. Pay particular attention to fields planted
after May 10.
Reports of heavy corn borer infestations continue,
some corn producers say this is the worst they have
seen in 20 years. Much of this is caused by
southwestern corn borer, but there is considerable
damage by European corn borer as well.
Southwestern corn borer is continuing to girdle stalks
just above the soil line and will continue to girdle additional
stalks through mid October. Producers need to
identify the most heavily infested fields and plan to
harvest them as soon as the moisture level of the grain
is acceptable. Pay particular attention to fields planted
after May 10.

 Powdery mildew, if left to develop unchecked, can
seriously impact crop yield. This is especially true for
fields where disease develops early (e.g., pre-flag leaf
emergence) and continues throughout the season.
When significant levels of powdery mildew are
detected for a field, impacted growers may consider
the option of disease management using either Tilt or
Quadris. Either fungicide will do the job, but if
applications are made before the early heading stages,
late-season disease control may be compromised. The
only option would be to make a second application
using Quadris. It is illegal to apply more that 4 fl oz/A
of Tilt to a crop, but the Quadris label does provide for
two applications. Excessive cost would, however,
assures that this option is not a viable one.
Powdery mildew, if left to develop unchecked, can
seriously impact crop yield. This is especially true for
fields where disease develops early (e.g., pre-flag leaf
emergence) and continues throughout the season.
When significant levels of powdery mildew are
detected for a field, impacted growers may consider
the option of disease management using either Tilt or
Quadris. Either fungicide will do the job, but if
applications are made before the early heading stages,
late-season disease control may be compromised. The
only option would be to make a second application
using Quadris. It is illegal to apply more that 4 fl oz/A
of Tilt to a crop, but the Quadris label does provide for
two applications. Excessive cost would, however,
assures that this option is not a viable one.

 Boxelder bugs are common insects that feed on sap
from leaves, twigs, and seeds of boxelders, as well
other members of the maple family. Large numbers of
them can be seen on tree trunks, or branches, or
sunning themselves on the south or west sides of
buildings in the spring and fall. These harmless
accidental invaders may be a temporary nuisance as
they move into and out of sheltered overwintering
sites in the fall and spring.
Boxelder bugs are common insects that feed on sap
from leaves, twigs, and seeds of boxelders, as well
other members of the maple family. Large numbers of
them can be seen on tree trunks, or branches, or
sunning themselves on the south or west sides of
buildings in the spring and fall. These harmless
accidental invaders may be a temporary nuisance as
they move into and out of sheltered overwintering
sites in the fall and spring.

 According to a statewide poll of Kentucky
householders, 93% expressed concern over finding
insects inside their home. More than half indicated
that a single cockroach, cricket, or spider would
prompt them to use a can of bug spray or call an
exterminator. What many people do not realize,
however, is that most pests discovered indoors have
flown or crawled in from outdoors.
According to a statewide poll of Kentucky
householders, 93% expressed concern over finding
insects inside their home. More than half indicated
that a single cockroach, cricket, or spider would
prompt them to use a can of bug spray or call an
exterminator. What many people do not realize,
however, is that most pests discovered indoors have
flown or crawled in from outdoors.


