

ANTS will become more active;
CORN ROOTWORM BEETLES will be emerging over the next two to three weeks;
encounters with CHIGGERS and TICKS will increase.


For the second growing season, the UK College of Agriculture has a fully functional web-based consulting system for rapid communication about real-time plant-related diseases and other problems. This system allows county Extension agents to upload digital images and key information in order to obtain an expert consultation.
New cases are triaged several times each day by UK plant diagnosticians, who either respond directly or assign the case to the appropriate Extension specialist. One of the unique features of this system is its capacity to invite experts from outside UK to consult on a particular case without compromising the overall security of the system: the consultant can only study and respond to the case on which s/he is invited to comment. Although every county Extension office can use the Digital Consulting System (DCS), at least one county office in every Extension District is equipped with a dissecting microscope and digital camera for transmission of images magnified up to 45X.
The relevance of this to agricultural biosecurity is that the DCS provides a rapid and secure means of communication between agents, specialists and national experts on plant disease issues that may be of regulatory significance. Time is of the essence when a new disease is introduced--whether by natural means or terrorist action--because efforts to contain and eradicate an introduced disease depend to a large degree on how quickly the infestation is detected.
For most plant disease problems, this system does not substitute for laboratory-based plant disease diagnosis, but it greatly increases communication abilities. In addition to increasing speed of communication, the DCS helps reduce the risk of accidental spread of a newly introduced disease. For example, if DCS consultation indicates that a disease problem may be new and of regulatory significance, Extension agents can be informed in advance about special handling precautions when preparing and submitting live plant samples.


 Nearly all the burley crop in KY has been set, and we've seen only a few cases of black shank thus far. By the end of June last year, drought prevailed in many parts of the Commonwealth and we saw a big jump in the number of cases of black shank being reported around that time. Many growers learned a hard lesson about black shank last year and are taking the necessary precautions against black shank: crop rotation, planting resistant varieties, and applying mefenoxam (Ridomil Gold or Ultra Flourish). Many growers put out mefenoxam prior to, or just after, planting and others have waited.
Nearly all the burley crop in KY has been set, and we've seen only a few cases of black shank thus far. By the end of June last year, drought prevailed in many parts of the Commonwealth and we saw a big jump in the number of cases of black shank being reported around that time. Many growers learned a hard lesson about black shank last year and are taking the necessary precautions against black shank: crop rotation, planting resistant varieties, and applying mefenoxam (Ridomil Gold or Ultra Flourish). Many growers put out mefenoxam prior to, or just after, planting and others have waited.
Fungicide options for black shank are available after transplanting. Ridomil Gold EC and Ultra Flourish, can be applied at first cultivation (if applicable) and at layby. The rate for Ridomil Gold is 1 pt/A (per treatment), while Ultra Flourish, which contains half the amount of mefenoxam found in Ridomil Gold, is labeled at 1 qt/A (per treatment). Applications should be directed at the soil and stems of plants. Mefenoxam needs adequate moisture in soil for maximum efficacy and should be incorporated after application with 1 inch of water if soils are dry. The fungicide should be incorporated as quickly after application as possible. Secondary spread of black shank is considerably less likely in a drought than in rainy weather; however, heavy rains or irrigation could result in heavy losses to black shank in fields with even low levels of disease, so mefenoxam should be applied in advance of anticipated moisture events. The black shank pathogen can be moved easily on equipment and feet! Growers need to sanitize properly when moving between infested and clean fields.
I have gotten a few inquiries about using Quadris to manage black shank on burley and dark tobacco. Although this is a practice that is gaining acceptance in some parts of the region, it is not recommended. Neither the Section 18 exemption for Quadris nor the pending Section 3 (full) label list black shank as a target disease. Does Quadris have activity against black shank? Quadris has been evaluated at the university level in several states, including KY, and results from these trials are not encouraging. Suppression of black shank has been erratic, and mefenoxam has been found superior to Quadris in most cases. So, in the end, growers should stick with Ridomil Gold or Ultra Flourish as the best fungicide options to manage black shank.
 Quadris has been granted an exemption from its existing label under Section 18 of FIFRA. The exemption covers two diseases of tobacco - frogeye leaf spot and target spot - and is valid from June 9, 2006 until October 15, 2006. Each grower must have a copy of the Section 18 in his or her possession at the time of application. A full (Section 3) label is expected sometime this summer that will include frogeye leaf spot, target spot, and blue mold.
Quadris has been granted an exemption from its existing label under Section 18 of FIFRA. The exemption covers two diseases of tobacco - frogeye leaf spot and target spot - and is valid from June 9, 2006 until October 15, 2006. Each grower must have a copy of the Section 18 in his or her possession at the time of application. A full (Section 3) label is expected sometime this summer that will include frogeye leaf spot, target spot, and blue mold.
Quadris should be applied only when conditions favor development of frogeye leaf spot or target spot. Frogeye is favored by warm, humid weather; target spot is more likely to occur during periods of moderate temperatures and high humidity or moisture (similar to conditions that favor blue mold). We encourage growers and agents to scout crops and report any frogeye or target spot to us as quickly as possible so that we can alert the tobacco community and begin timely application of Quadris.
The recommended rate of application is 8 oz/A, for a maximum of 32 oz/A or 4 applications during the season. Quadris can be applied up to the day of harvest, but only one application of the fungicide is permitted in the 30 days prior to harvest. According to my sources, Quadris retails for around $320 per gallon, making the cost of an application roughly $20 per acre. Last year, I learned that some growers were told by their chemical dealers that 6 fl oz/A of Quadris was adequate to achieve good disease control. Using Quadris at less-than-recommended rates is legal, but control under high disease pressure might be unacceptable. What's more, the manufacturer will not be liable for control failures where reduced rates are used. Applying the proper 8 fl oz/A rate costs about $5/A more than the 6 fl oz/A application, but the extra cost could well be worth it considering the explosive nature of tobacco diseases and the high value of the crop.
Quadris is not a systemic fungicide, but does have "translaminar" activity. This means that active ingredient will migrate from treated leaf surfaces to the untreated sides after a brief period of time, and this could provide some curative activity. That being said, Quadris should be applied preventively for maximum suppression of disease. This means putting out the material at the earliest signs of disease at the latest. Applying Quadris to heavily infected crops can lead to development of resistance in pathogen populations and a loss of this important tool for disease management.
 Blue mold was found in Carter Co. around June 13 on direct-seeded transplants in a float bed. Some tobacco from this particular float bed was set, and active blue mold may be present in the field. We have reason to believe that the Carter Co. outbreak is associated with plug plants purchased from Florida.
Blue mold was found in Carter Co. around June 13 on direct-seeded transplants in a float bed. Some tobacco from this particular float bed was set, and active blue mold may be present in the field. We have reason to believe that the Carter Co. outbreak is associated with plug plants purchased from Florida.
At the moment, I am urging our growers to keep up preventive applications of fungicide to be on the safe side. Let me know also if you find blue mold or suspect it in your area, and check the Kentucky Tobacco Disease Information page for regular updates on blue mold and other diseases
For the latest blue mold status and other tobacco disease information, check the KY Blue Mold Warning System online.
![]() http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm
http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm

For more information about tobacco pests, visit "Insect Management Recommendations".

![]() I have received a few calls and samples concerning the presence of soybean aphids in our soybean fields. Several people have submitted small yellow insects thinking they might be soybean aphids. They were not. They are a bright yellow thrips. As of this writing, no sample submitted to the Insect ID lab contained soybean aphids, no samples from the 16 sentinel plots contained soybean aphid, nor have we captured any in the aphid suction trap in Princeton, KY.
I have received a few calls and samples concerning the presence of soybean aphids in our soybean fields. Several people have submitted small yellow insects thinking they might be soybean aphids. They were not. They are a bright yellow thrips. As of this writing, no sample submitted to the Insect ID lab contained soybean aphids, no samples from the 16 sentinel plots contained soybean aphid, nor have we captured any in the aphid suction trap in Princeton, KY.
It is very easy for the casual or first time observer to mistake a young soybean thrips for soybean aphid. However, once you have seen each of them they are actually very easy to tell apart.
There may be more than one type of thrips on soybeans. Thrips have two larval stages which range in size from 0.6 to 1.0 mm (0.023 to 0.039 inch) long. Larvae are white when newly hatched, then gradually turn yellow with age. The soybean thrips larvae eventually turn orange with some red pigmentation, though the body sometimes has a greenish tint due to ingested chlorophyll.
Adult thrips are 1.25 mm (0.049 inch) or less in length and have two pairs of long, narrow wings fringed with long hairs. Adult soybean thrips has a yellow body with a dark blotch on the back behind the head and two distinct cross-bands on the forewings.
Generally Note-
Aphids:
Thrips:
Go to the KY-IPM Soybean Aphid page to see a color photo of the aphid. http://www.uky.edu/Ag/IPMPrinceton/SoybeanAphid/sbaphidindex.htm
To see other color photos look at the national soybean aphid pest alert at: http://www.ncpmc.org/alerts/soybeanaphid_alert.pdf
Color photos of thrips: http://www.gaipm.org/top50/thrips.html http://www.mda.state.mn.us/biocon/plantscape/thrips.htm
See soybean aphid and thrips on the same page http://www.ent.iastate.edu/Ipm/Icm/2004/7-12-2004/thrips.html
![]() As I write this article (June 19, 2006) the soybean rust risk in Kentucky soybean is very low. I do not foresee a change in this situation at least through early July. During the next 30 days, many acres of Kentucky soybean will be in the early reproductive stages (R2 - full flowering to R3 - early pod development). This is the target period for "plant health" application of fungicides. These treatments are being promoted by a couple of major fungicide manufacturers as a means of promoting enhanced plant health, leading to higher crop yields. Suggested treatments involve strobilurin-containing fungicides, such as pyraclostrobin (e.g., Headline) or azoxystrobin (e.g., Quadris).
As I write this article (June 19, 2006) the soybean rust risk in Kentucky soybean is very low. I do not foresee a change in this situation at least through early July. During the next 30 days, many acres of Kentucky soybean will be in the early reproductive stages (R2 - full flowering to R3 - early pod development). This is the target period for "plant health" application of fungicides. These treatments are being promoted by a couple of major fungicide manufacturers as a means of promoting enhanced plant health, leading to higher crop yields. Suggested treatments involve strobilurin-containing fungicides, such as pyraclostrobin (e.g., Headline) or azoxystrobin (e.g., Quadris).
In the absence of soybean rust and significant risk of infection by the rust pathogen, the decision to apply a "plant health" fungicide application is not as straight forward as some might believe. There is not general agreement among university and industry scientists regarding the potential for plant health applications to result in economical benefits to producers. By economical, I mean that the $ return per acre exceeds the $ invested in fungicide/application costs.
During 2003-05, the Kentucky Soybean Promotion Board invested a significant amount of your check-off dollars to determine the potential for "plant health" fungicide applications to result in an economic benefit to producers. Funds were given to the University of Kentucky and WheatTech, Inc. to facilitate independent study of this question. Tests conducted by the University of Kentucky involved both large and small plot studies during 2003-05. WheatTech studied the question in a small plot tests during 2004-05 tests. The results of tests were mixed, but generally, applications of fungicides to soybean, in the absence of specific foliar fungal diseases, frequently did not produce a statistically significant (i.e., economical) yield response. Specifically, over the three years of study, significant yield results were encountered in six (6) of twenty-four (24) experiments (25.0% response rate). A similar rate of statistically significant treatment responses (26.9%) was seen in 62 replicated experiments conducted throughout the Midwest and Northeast during 2005. These experiments involved both large and small plot tests. All of the experiments referenced so far involved replication of treatments, which gave scientists the ability to statistically analyze treatment results. The ability to statistically analyze treatment results may seem more "ivory tower" than practical, but in fact, just the opposite is true. From a practical perspective, the only way to know, with any degree of certainty, if one treatment produced a different result than another treatment is to subject results to the rigor of accepted statistical procedures. Anything less results in one guessing if one treatment really had an impact, or if apparent differences were merely the result of chance and/or inherent experiment variability.
In contrast to the above, results of "plant health" fungicide applications based on a large number of strip plot, side by side comparisons, in grower fields have been much more favorable to potential yield benefits. For example, 162 side by side comparisons, summarized by BASF from Indiana and Ohio in 2005, showed an average yield response to "plant health" fungicide applications of 5.6 bu/ac (3 - 4 bu/ac is considered to be break even, depending on the fungicide and application cost, and the price received per bushel of soybean). Comparisons summarized from 192 fields across the Midsouth in 2005, including Kentucky, were even more favorable with an average yield response of 8.5bu/ac. It is my understanding is that Syngenta can produce similar results from their on-farm trials over the last few years. There are many inherent problems with non-replicated, side by side, strip plot comparisons. One major concern is that results typically do not include detailed information on disease activity. Thus, one cannot know if a disease was the reason for the success or failure of a treatment. In addition, treatments in a field cannot be statistically analyzed because there is no in-field treatment replication. Still, large numbers of strip trials with similar results have meaning and value, albeit serious questions still exist about the data generated.
What is one to make of the apparent discrepancy between the results of 84 replicated experiments, conducted by numerous scientists, and the results of non-replicated, but numerous, on-farm treatment comparisons conducted by industry? To be honest, I am not sure. Certainly, the great disparity in results should be a "red flag" to anyone considering making applications of foliar fungicides to soybean in the absence of specific foliar fungal target diseases. On the other hand, very high treatment yields in many strip plot comparisons (and some replicated experiments) over the last few years make it clear that something significant is going on. I have seen enough positive results to conclude that "plant health" applications of fungicides to soybean cannot not be discarded, offhand. Yet, I have also seen enough negative results to know that there are many, many scenarios where "plant health" fungicide applications simply will not pay for themselves.
At this point, I would simply caution producers who are considering making fungicide applications for plant health benefits, to make those applications with the awareness that a significant economic response is not assured. At this point, we (university and industry scientists) do not have enough information to be able to recommend which fields/situations have a high probability of an economic response and which fields have a low probability of response. In addition, applications of any pesticide in the absence of a specific target pest(s) is contrary to integrated pest management principles. Thus, there may be unintended, long term implications (environmental, regulatory, and/or sociological) associated with applying fungicides to soybean in the absence of specific target pests. One consequence could be the suppression of native fungi that keep insect populations in check, naturally. There is already some evidence that indiscriminate use of fungicides in soybean has resulted in increased insect/mite activity in some states. Another consequence could be stricter future pesticide laws and regulations. The point is we must be very cautious when we get into the realm of applying fungicides (or any pesticide) to large acreages of soybean (or any crop), even if there is a good rationale for doing so. In this article, I am making it known that the rationale for making "plant health" fungicide applications to soybean is not firmly in favor of making the applications. Thus, caution is advised at numerous levels.
For more information about soybean pests, visit
"Insect Management Recommendations".


Commercial apple growers with unsprayed trees and backyard apple growers throughout Kentucky are observing bright orange spots on apple tree leaves. These orange spots represent symptoms and signs of cedar-apple rust disease. Cedar rust diseases are widespread this year because of wet spring weather favored disease development.
Symptoms. Cedar-apple rust, caused by Gymnosporangium juniperi-virginianae is the most common and important of the rust diseases on apple. On apple leaves, small, pale yellow spots (sites of rust fungus infection) appear on the upper surface in mid to late spring. These yellow spots turn a glistening orange color and gradually enlarge (up to �-inch in diameter, depending on the apple variety and the number of spots per leaf) and will become bright yellow-orange frequently surrounded by a reddish border. As the spots enlarge, black dots (a mass of fungal fruiting bodies called pycnia) develop in the centers and this is being observed now. Soon the fungus will grow through to the lower leaf surface, where yellow spots also appear, and the tissue becomes noticeably thickened. In early summer, clusters of small orange-yellow, tubular fruiting bodies (aecia) will project downward from these lower surface spots. As the "tubes" mature, they split toward the base in narrow strips and curl back on themselves to form "cups" within which a mass of light brown spores is revealed. Heavily infected leaves may turn yellow and drop, especially as the tree becomes stressed for water.
Apple fruits infected with cedar-apple rust develop spots appearing near the blossom end. They are yellow-orange in color, like the leaf spots, but are much larger (up to �-inch or more in diameter), and are surrounded by a dark green zone on the otherwise light green fruit. The tubular aecia frequently fail to develop but when present, they are usually found in a circle surrounding the black dots (pycnia) which form on a raised, roughened cushion of tissue. Tissue below the spots turns somewhat corky, but remains alive. Infected fruits frequently become deformed and may drop prematurely. Cedar-apple rust normally does not affect apple twigs.
Cedars (junipers) are alternate hosts for the cedar rusts, but obvious rust disease symptoms are not now evident on them. The cedar-apple rust fungus forms light brown to reddish or chocolate brown galls in the leaf axils of infected Juniperus species. These galls are usually rounded and range from pea-sized to 2 inches in diameter. As galls mature, the flesh becomes corky and the surface becomes pitted with circular depressions. In spring, following rainy periods, slimy, yellow-orange tendrils, or "spore horns," up to 2 inches long swell and protrude from these depressions. A gall may produce many spore horns, which cause it to resemble orange-colored blossoms from a distance. Severely rusted Juniperus can be very conspicuous and many were this past spring.
Disease cycle. Aeciospores being produced now or very soon in the tubular aecia on diseased leaves are blown to cedar during the summer where they germinate, infect, and cause small pea-sized galls to form on the twigs. The fungus over-winters in these galls which continue to grow and enlarge during the following year. The fungus survives a second winter within the gall, then begins producing its long, orange spore horns the next spring, about the time the apple buds are in the pink to early bloom stage. These spore horns are actually columns of fungal spores (teliospores), each of which can germinate under moist conditions to form four new spores (basidiospores). Basidiospores are then carried to apple trees by wind currents where they can germinate and cause infection during relatively short periods of wetness (about six or seven hours for moderate levels of infection when temperatures are in the 50s and 60s). The danger of infection usually ends about 30 days after apple bloom when the fungus terminates production of basidiospores on cedars and the majority of apple leaves have aged to the point where they are no longer susceptible. Spots begin forming on the upper leaf surface about 10 to 14 days after infection occurs, and aecia form on the underside of infected leaves several weeks later. Spores produced within the aecia are then blown to nearby cedars and junipers, completing the cycle that began two years earlier. Thus, during the relatively wet summer of 2004, high levels of juniper infection may have occurred and resulted in more juniper galls producing inoculum for infections this spring of 2006.
Cedar-apple rust disease management.

Grape berry moth is one of the key pests of grapes that can cause significant losses in some years. The larvae of this insect damages commercial vineyards by feeding on the grape blossoms and berries. Infested berries may appear shriveled with fine webbing. Damage by grape berry moth may increase mold, rots and numbers of fruit flies. While grape berry moth larvae may only damage a few berries in a cluster, it is impractical for growers to remove damaged berries and webbing from clusters. Hosts include wild and cultivated grapes.
Adult moths emerge in late May and June to lay eggs of the first generation singly on fruit stems near bloom time. Eggs hatch in about 5 days. Under a flimsy web, the larvae feed for about 21 days on the blossoms and young fruit. In mid July, larvae move to leaves where they make a semi-circular slit, fold the flap over themselves and pupate. Adult moths emerge from the pupae in 10 to 15 days. Moths begin laying eggs for the next generation after 4 to 5 days. There may be 2 or 3 generations per year. Larvae of the second and third generations enter berries and feed within, passing from one berry to another under protection of webbing. Some of the cocoons of the second or third generations fall to the ground where they overwinter. Webbing over blossoms and berries, and leaf flap cocoons are indicative of grape berry moth. In winter, the cocoons may be found in leaf litter under the vines.
First generation control is important as this limits the number of eggs for the second generation. To control the first generation, typically two applications are applied about 10-days apart. Insecticidal control of second generation is more difficult due to an extended flight period of moths as well as the difficulty of getting adequate spray coverage inside the cluster as berry size increases. Sprays for the second generation should be timed for the veraison stage of development. Pheromone traps are available to monitor for adult moth activity and enhance timing of insecticides for grape berry moth control.
Recent studies in some states have shown mating disruption with synthetic pheromones to be an effective alternative in situations where there is no immigration of moths from outside sources. Mating disruption relies on releasing enough of the pheromone in the vineyard so that males cannot find female moths. Eggs laid by unmated females fail to hatch. Various systems are commercially available for mating disruption of grape berry moth. Minimum vineyard size may vary with the type of mating disruption used.


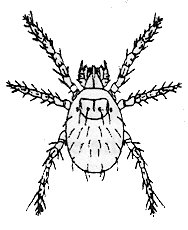 Chigger bites, a sure sign of summer, can be the "souvenir" of a blackberry-picking trip, working, or playing in overgrown brushy or grassy areas. Chiggers usually feed where clothing fits tightly against the skin - waistbands, etc. Digestive juices used by the mites to dissolve skin cells cause bite sites to become red and swollen. Later, angry red welts will form that ooze and itch intensely for several days, even though the chiggers are long gone. As if that were not enough, bite sites can become infected if they are scratched frequently with dirty fingernails. The tips below can be useful in minimizing problems with ticks and chiggers - which hang out in the same settings.
Chigger bites, a sure sign of summer, can be the "souvenir" of a blackberry-picking trip, working, or playing in overgrown brushy or grassy areas. Chiggers usually feed where clothing fits tightly against the skin - waistbands, etc. Digestive juices used by the mites to dissolve skin cells cause bite sites to become red and swollen. Later, angry red welts will form that ooze and itch intensely for several days, even though the chiggers are long gone. As if that were not enough, bite sites can become infected if they are scratched frequently with dirty fingernails. The tips below can be useful in minimizing problems with ticks and chiggers - which hang out in the same settings.
Wear long pants tucked into boots or socks to keep chiggers and ticks on the outside of your clothing. Wear loose fitting, light colored clothing and avoiding sitting or lying directly on the ground.
Avoid walking through overgrown fields and brush, especially from July through early September. Walk in the center of trails to avoid tall vegetation where chiggers and ticks congregate.
Use an insect/ tick repellent. Products containing diethyl toluamide (DEET) or permethrin (clothing only) are most effective. Be sure to read and follow directions for use on the container. A hot, soapy shower immediately after coming indoors will remove chiggers and ticks that have not yet attached.
Chigger / Tick management
Control of chigger and ticks in large yards, parks, camps, picnic sites, and other recreational areas is often impractical. However, chiggers in play and picnic areas and around trails can be reduced by vegetation management. Regular mowing and brush removal creates a less favorable habitat for chiggers and the rodents and other small animals on which they feed. This is the way to a long-term solution.
Insecticide sprays may provide some temporary reduction of chiggers. They are most effective when directed into areas where chiggers and their animal hosts are likely to frequent. Options include bifenthrin (Ortho Lawn Insect Killer), carbaryl, (Sevin), cyfluthrin (Bayer Advanced Lawn and Garden Multi-Insect Killer, and any of a number of products containing permethrin. Be sure to read the product label carefully to be sure the site you are planning to treat is on the label. Also, look for specific instructions for applications against chiggers that can increase control.
How do all those blood-feeding creatures find us? Vision, warmth, and odor are clues that many biting pests use to find a blood meal. The importance of each of these factors, and many others but here are some basics: Vision can play a large role in long distance recognition and attraction. For example, day-feeding mosquitoes tend to respond to movement, especially of people in dark clothing. Differences in body temperature may be the reason that some mosquitoes feed in relatively specific sites such as the ankles or head. Odors come into play as the mosquito gets very close. This is a very complex area with skin bacteria joining into the mix.
Modified in part from Mosquitoes and Mosquito Repellents: A Clinician's Guide by Mark S. Fradin, MD. In Annals of Internal Medicine, 1 June 1998. 128:931-940. http://www.acponline.org/journals/annals/01jun98/mosquito.htm
DEET has been the standard "bug" repellent for about 50 years and is still recognized as providing effective, long term protection against mosquitoes and many other blood-feeding arthropods. It is available in a wide range of products with varying concentrations of the active ingredient. However, some object to deet's odor or the oily feeling that it leaves on the skin. Deet can irritate eyes and mucous membranes making applications to the face difficult and there are concerns about applications to children.
The CDC (Centers for Disease Control) has added picaridin and oil of lemon eucalyptus as effective repellents. They have been shown to provide protection comparable to products containing 10% deet. Target pests include biting flies, mosquitoes, ticks, and chiggers.


 Adult Japanese beetles and masked chafers have begun to emerge. As is usually the case, it is difficult to predict how serious a problem these pests will be this year. Both Japanese beetles and masked chafers lay eggs in moist soil under turf. Their grubs then feed on turfgrass roots.
Adult Japanese beetles and masked chafers have begun to emerge. As is usually the case, it is difficult to predict how serious a problem these pests will be this year. Both Japanese beetles and masked chafers lay eggs in moist soil under turf. Their grubs then feed on turfgrass roots.
Japanese Beetles (Adults) - Detailed information on this pest can be found in ENT-5, Japanese Beetles in the Urban Landscape. Options for protecting landscape plants from foliage feeding adults are as follows:
Plant Selection- The best way to avoid perennial battles with adult Japanese beetles is to select plant material that is less preferred. Publication ENT-5 lists species and cultivars of trees and shrubs that are less likely to be attacked by beetles.
Hand Picking and Exclusion- For smaller plants, it may be practical simply to remove the beetles by hand. Volatile odors released from beetle-damaged leaves attract more beetles. By not allowing Japanese beetles to accumulate, plants will be less attractive to other beetles. One of the easiest ways to remove beetles from small plants is to shake them off early in the morning when the insects are sluggish. The beetles may be killed by shaking them into a bucket of soapy water. Highly valued plants such as roses can be protected by covering them with cheesecloth or other fine netting during peak beetle activity (usually late June to mid-July).
Insecticides- Carbaryl (Sevin) and several pyrethorid products such as bifenthrin (TalstarOne), cyfluthrin (Tempo, Bayer Advanced Lawn& Garden Multi-Insect Killer), deltamethrin (Deltaguard), lambda cyhalothrin (Scimitar, Spectracide Triazicide), and permethrin are labeled for control of adult Japanese beetles. The pyrethroids generally gave 2-3 weeks protection of plant foliage while carbaryl gave 1-2 weeks protection. Foliage and flowers should be thoroughly treated. The application may need to be repeated to prevent re-infestation during the adult flight period. Follow label directions and avoid spraying under windy conditions. For gardeners seeking a botanical alternative, Neem (e.g., Azatrol, Neem-Away from Gardens Alive), or Pyola (pyrethrins in canola oil) provided about 3-4 days deterrence of Japanese beetle feeding. Insecticidal soap, extracts of garlic, hot pepper, or orange peels, and companion planting, however, were found to be non-effective.
White Grubs - There is no reliable way to predict whether any given year will be a bad one for white grubs - the immature, turf-feeding stages of Japanese beetles, masked chafers, and certain other beetles. Moreover, since grub infestations tend to be localized and sporadic, only small percentages (< 10 percent) of Kentucky lawns require treatment, even in bad years for grubs.
Indicators of Infestation- White grubs and their resultant damage are not usually evident until August or September. Although sampling the turf is the only way to confirm that grubs are present, certain factors may indicate an increased risk of infestation later in the season. If your turf has a history of serious grub problems, there is a greater chance that adult beetles will return and re-infest the same areas. Sites with large numbers of adult beetles in June and July are more likely to have grubs in late summer. Early warning signs include swarms of brown, �-inch long masked chafer beetles skimming over the turf at dusk, or green June beetles buzz-bombing the turf by day in search of mates and egg-laying sites. Masked chafer and May beetle adults are also attracted to porch and streetlights at night. Heavy infestations of adult Japanese beetles feeding in the area might also foretell subsequent problems with grubs of that species.
Rainfall and soil moisture are critical factors affecting the extent of grub damage during a season. Frequent irrigation in June and July may attract egg-laying female beetles to the turf, especially if surrounding areas are dry. High soil moisture also increases egg survival. If lawns are irrigated during periods of dryness in June and July, be especially alert for signs of grubs later in the summer. Conversely, adequate soil moisture in August and September (when grubs are actively feeding) can help to hide root injury. Irrigated turf can sometimes tolerate 20 or more grubs per square foot before showing signs of injury.
Treatment Strategies - Two different strategies are available for controlling white grubs with insecticides: preventive and curative. Each approach has its own merits and limitations. With preventive control, the insecticide is applied as insurance, before a potential grub problem develops. Consequently, preventive control is best suited for high-risk sites with a history of grub problems, or turf sites where heavy beetle activity is noted.
Preventive control requires the use of insecticides with long residual activity in soil. Look for products containing the active ingredient imidacloprid (e.g. Merit�, Bayer Advanced� Season-Long Grub Control) or halofenozide (e.g. Mach 2�). Both of those ingredients have sufficient soil persistence to apply anytime from early June to mid-July and still control young grubs hatching from eggs from mid-July to early August. The optimum treatment period for these products is mid-June to mid-July.
Preventive treatments afford greater flexibility in application timing, and are easier to schedule and implement than are curative treatments. They often afford greater peace of mind to golf superintendents and lawn service companies because potential damage is avoided or minimized. The main drawback of preventive grub control is that the decision to treat must be made before knowing the extent of infestation. Grub outbreaks tend to be localized and sporadic and only a small percentage of lawns require treatment in a given year. Thus, preventive control often results in areas being treated unnecessarily. Good record keeping and observation will help in pinpointing grub-prone areas, which are the most logical candidates for preventive applications.
With curative control, treatment is applied in late summer - typically August or September - after the eggs have hatched and grubs are present. Ideally, the decision to treat is based on site inspection and sampling or past history of infestation. Since white grub infestations tend to be localized, the entire lawn often will not need to be treated. Grub "hot spots," which can be confirmed by sampling, are most likely to be full sun, south or west-facing slopes, lawns seeded with Kentucky bluegrass, lawns that were heavily irrigated during June and July, and turf areas that were damaged by grubs in previous years.
Proper timing of curative grub treatments can be tricky. Insecticides applied before early August may degrade before the eggs have hatched, whereas if the product is applied in late August or September, the grubs will be large and harder to kill and severe damage to turf may have already occurred. Granular formulations containing the active ingredient trichlorfon (e.g. Dylox, Bayer Advanced 24-hour Grub Control) are the fastest-acting, most effective insecticides for curative grub control. There is little benefit in applying a short-lived, curative-type product for white grubs in June or July.


Agronomic samples received in the PDDL this past week included Lepto leaf spot, boron deficiency, spring black stem, and potato leafhopper injury on alfalfa; zinc deficiency and maize dwarf mosaic virus on corn; black root rot, tomato spotted wilt virus, manganese toxicity, Pythium root rot, target spot, Fusarium wilt, transplant shock and chemical injury on tobacco.
On fruit and vegetable samples, we diagnosed acid soil problems on blueberry; orange rust on blackberry; cedar-apple rust and frogeye leaf spot on apple; Botryosphaeria canker on cherry; fire blight on pear; scab on apricot; anthracnose on cucumber; Choanephora rot on squash; Rhizoctonia stem rot on bean; potyvirus on garlic; tomato spotted wilt virus, bacterial canker, and early blight on tomato; and Pythium root rot on watermelon.
Ornamental and turf samples included bacterial soft rot on iris; cedar-quince rust on
hawthorn; fire blight and scab on crabapple; bacterial leaf spot on hydrangea;
bacterial blights on mulberry and walnut; Guignardia leaf blotch on horsechestnut;
anthracnose on oak, maple, ash, and dogwood; leaf blister and powdery mildew on
oak; chestnut blight on American chestnut; brown patch on fescue; Pythium blight
on bentgrass; and rust on bluegrass.


UKREC-Princeton, KY, June 9-16, 2006| True Armyworm
| 3
| Corn Earworm
| 6
| European Corn Borer
| 0
| Southwestern Corn Borer
| 3
| Fall Armyworm
| 1
| Black Cutworm
| 6
| | |
View Princeton trap counts for the entire 2006 season at - http://www.uky.edu/Ag/IPMPrinceton/Counts/2006trapsfp.htm
Fulton County trap counts are available at -http://ces.ca.uky.edu/fulton/anr/Insect%20Counts.htm
For information on trap counts in southern Illinois visit the Hines Report at - http://www.ipm.uiuc.edu/pubs/hines_report/comments.html The Hines Report is posted weekly by Ron Hines, Senior Research Specialist, at the University of Illinois Dixon Springs Agricultural Center.
NOTE: Trade names are used to simplify the information presented in this newsletter. No endorsement by the Cooperative Extension Service is intended, nor is criticism implied of similar products that are not named.
Lee Townsend
Extension Entomologist