winged TOBACCO APHIDS and TOBACCO BUDWORMS begin to appear in earliest set fields;
check for standing water that could be a breeding site for MOSQUITOES.


JAPANESE BEETLES starting to emerge in central Kentucky;
winged TOBACCO APHIDS and TOBACCO BUDWORMS begin to appear in earliest set fields;
check for standing water that could be a breeding site for MOSQUITOES.


 Blue mold has not been reported in the United States as of June 13. Tropical storm Arlene, which moved
through our area this weekend, brought heavy rains to some locations. Trajectory models from the
National Plant Disease Forecasting Center indicate that any blue mold spores present in traditional
source areas such as Pinar del Rio, Cuba and Uvalde, Texas were likely moved well west of U.S.
production areas by the storm. Current forecasts indicate that we are heading into a period of
drier weather; however, growers still need to scout fields regularly for symptoms of blue mold and
other diseases.
Blue mold has not been reported in the United States as of June 13. Tropical storm Arlene, which moved
through our area this weekend, brought heavy rains to some locations. Trajectory models from the
National Plant Disease Forecasting Center indicate that any blue mold spores present in traditional
source areas such as Pinar del Rio, Cuba and Uvalde, Texas were likely moved well west of U.S.
production areas by the storm. Current forecasts indicate that we are heading into a period of
drier weather; however, growers still need to scout fields regularly for symptoms of blue mold and
other diseases.
 The nutrient-rich water standing in tobacco float beds can breed thousands of mosquitoes in a very short time. Stagnant water mosquitoes, such as the house mosquito, lay rafts of eggs in the surface of stagnant water. Development from egg to adult takes from 8 to 12 days. The black plastic bed liners warm the water so the mosquito larvae (wriggles) and pupae (tumblers) will develop quickly. A few minutes spent looking carefully at the water will allow you to see the active wrigglers and tumblers as they come to the surface for air.
Dumping the water will kill the developing mosquitoes. But folded plastic left outside will have pockets that will collect water and allow continued mosquito breeding. If some beds are needed for backup plants and immature mosquitoes are living in the water, then products containing Bti (Bacillus thuringiensis israeliensis) can be applied to kill them. These products only kill mosquito larvae; they will not control pupae or adults.
House mosquitoes are members of the genus Culex, a group that contains important vectors of West Nile and other encephalitis viruses. They feed primarily on birds but also feed on mammals and will enter structure and feed on humans.
The nutrient-rich water standing in tobacco float beds can breed thousands of mosquitoes in a very short time. Stagnant water mosquitoes, such as the house mosquito, lay rafts of eggs in the surface of stagnant water. Development from egg to adult takes from 8 to 12 days. The black plastic bed liners warm the water so the mosquito larvae (wriggles) and pupae (tumblers) will develop quickly. A few minutes spent looking carefully at the water will allow you to see the active wrigglers and tumblers as they come to the surface for air.
Dumping the water will kill the developing mosquitoes. But folded plastic left outside will have pockets that will collect water and allow continued mosquito breeding. If some beds are needed for backup plants and immature mosquitoes are living in the water, then products containing Bti (Bacillus thuringiensis israeliensis) can be applied to kill them. These products only kill mosquito larvae; they will not control pupae or adults.
House mosquitoes are members of the genus Culex, a group that contains important vectors of West Nile and other encephalitis viruses. They feed primarily on birds but also feed on mammals and will enter structure and feed on humans.
For the latest blue mold status and other tobacco disease information, check the KY Blue Mold Warning System online.
![]() http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm
http://www.uky.edu/Agriculture/kpn/kyblue/kyblue.htm

For more information about tobacco pests, visit "Insect Management Recommendations".

![]() Tropical storm/depression Arlene made its way across the panhandle of Florida, across parts of southern Louisiana, most of Georgia, Alabama, Mississippi, and Tennessee, and into parts of west Kentucky during June 10-12, 2005. The system moved over the southern Great Lakes regions and into the northeast U.S. on Monday, June 13.
Tropical storm/depression Arlene made its way across the panhandle of Florida, across parts of southern Louisiana, most of Georgia, Alabama, Mississippi, and Tennessee, and into parts of west Kentucky during June 10-12, 2005. The system moved over the southern Great Lakes regions and into the northeast U.S. on Monday, June 13.
The soybean rust model being maintained by USDA scientists indicates that significant soybean rust spore deposition MAY have occurred in a swath across the states affected by Arlene. However, a caveat is added that while model simulations indicate that spore movement into the southeastern U.S., with possible infection of susceptible hosts, may have occurred, the lack of knowledge of spore source areas and levels makes it impossible to assess the true threat of disease at this time. For example, it is unknown if Cuba is being affected by soybean rust and, if so, to what extent. Similarly, we have minimal information at this time on the extent of possible spore movement from other countries south of the U.S. This obviously has a tremendous bearing on how much spore movement may have occurred with Arlene.
Prior weather patterns existed that are predicted to result in soybean rust symptom expression in susceptible hosts in Florida, Georgia, southern South Carolina and south central Alabama this week. However, because of the latent period of 8-12 days between infection and symptom development under field conditions, the earliest we could expect to see soybean rust in Kentucky would be June 20-24. Of course, if no spores, or very low levels of spores, arrived here with the remnants of Arlene, it may be well into July or much later before we see any signs of soybean rust in Kentucky.
Right now, and until we have some indication that soybean rust has moved out of its current locations in central Florida and southwest Georgia, the prudent thing to do at this time is to wait, watch, and listen. Wait: now is NOT the time to spray fungicides! Watch: watch for any unusual occurrences in your soybean crop. Send us samples of anything that is suspicious. Instructions for packaging and submitting samples to the UK Plant Disease Diagnostic Laboratories can be found at the UK soybean rust website (www.uky.edu/soybeanrust). Listen to ag radio programs, read the most current updates and regularly visit the USDA soybean rust website for updates. I am placing commentary for Kentucky on that website on a regular basis (multiple times a week). Other extension specialists from neighboring states are doing likewise. I encourage you to read what my colleagues from other states are saying and compare it with what I am saying. There are unique aspects to each state, so do not expect everyone to say the same thing. Nonetheless, I think you will find some consistent trends, statements, and ideas that will give you some confidence that the system is working properly. State sentinel plot coordinators and specialists continue to meet by phone conference call each Monday in an attempt to stay up-to-date with what is going on across the south. Please understand that the level of cooperation and communication is extremely high.
I will pass on more information as it becomes available.
For more information about soybean pests, visit
"Insect Management Recommendations".


Kentucky fruit and vegetable growers may have noticed that phosphorous acid has been listed for management of several fruit diseases in recent editions of the commercial tree fruit and small fruit spray guides and management of several vegetable diseases on recent product labels. Some County Extension Agents and growers have been curious to know how they work. Parts of the following material was adapted from an insightful article written by Annemiek Schilder, Michigan State University Plant Pathologist, and was published in the Michigan State University Fruit Crop Advisory Team Alert, Vol. 20, No. 5, May 10, 2005.
Phosphorous acid fungicides. Recently, a number of new fungicides that have phosphorous acid as the active ingredient have come on the market. Other names sometimes used for this group are phosphonates or phosphites. Commercial products in this group may include ProPhyt�, Phostrol� and Agri-Fos�. Aliette� (fosetyl-Al), an older fungicide, is the prototype for this group of fungicides. However, the long-standing patent on Aliette� had prevented similar fungicides from being developed until recently. In Australia, where the patent did not apply, growers have been using the phosphorous acid fungicides for over a decade.
Phosphorous acid is not fertilizer. The term "phosphorous acid" should not be confused with phosphoric acid or phosphorus (P), a fertilizer component. In fertilizers, P is normally found in the form of phosphoric acid (H3PO4), which readily disassociates to release hydrogen phosphate (HPO42-) and dihydrogen phosphate (H2PO4 -). Both of these ions may be taken up by the plant and are mobile once inside the plant. Phosphorous acid is H3PO3. A single letter difference in the name of a chemical compound can make a major difference in its properties.
Phosphorous acid releases the phosphonate ion (HPO32-; also called phosphite) upon disassociation. Phosphonate is easily taken up and translocated inside the plant. Phosphorous acid does not get converted into phosphate, which is the primary source of P for plants. Because phosphorous acid and its derivatives do not get metabolized in plants, they are fairly stable and probably contribute little or nothing to P nutritional needs of the plants.
Some researchers have investigated the ability of phosphorous acid to act as a nutrient source for plant growth and found that P-deficiency symptoms developed with phosphorous acid as the sole source of P. This means that although phosphorous acid can control diseases, it is not a substitute for P fertilization. The inverse is also true: phosphate is an excellent source of P for plant growth, but is unable to control diseases other than improving the general health of the crop. So applying high amounts of P fertilizer will not work as a disease control measure.
Diseases managed with phosphorous acid. Researchers have found that phosphorous acid fungicides are especially effective against Oomycete pathogens, such as Phytophthora, Pythium, and downy mildews in a number of crops. Our fruit spray guides list Phytophthora collar rot and root rot of tree fruits, blueberries, and brambles; strawberry red stele and leather rot; and grape downy mildew as targets for phosphorous acid fungicides. Phosphorous acid is labeled for use on brassicas (broccoli, cabbage, cauliflower); cucurbits; edible legumes; Solanaceous crops (eggplant, pepper, potato, tomato, and tobacco); onions; and a number of leafy vegetables for management of diseases caused by Phytophthora, Pythium, Rhizoctonia, and Fusarium spp. Downy mildews of these crops are also listed on product labels. Materials containing phosphorous acid, in general, have performed best against Phytophthora blight and downy mildews. Preventive applications were more effective in reducing season-long severity of disease than those applied after disease onset.
Phosphorous acid has a direct and possibly an indirect effect on these pathogens. It inhibits a particular metabolic process (oxidative phosphorylation). In addition, some evidence suggests that phosphorous acid has an indirect effect by stimulating the plants natural defense response against pathogen attack. This probably explains the much broader spectrum of activity observed in many fungicide efficacy trials. In fruit crops, it has been found, for instance, that ProPhyt had efficacy against downy mildew, Phomopsis cane and leaf spot, and black rot (but not much against powdery mildew) in grapes. There is also evidence of activity of these compounds against anthracnose in blueberries.
The phosphonate ion is highly systemic and fairly stable in plants. The systemic activity allows them to be applied as foliar fungicides for prevention of Phytophthora and Pythium root rots. They may also display some curative activity. In general, applications every 14 days seem to be effective in grapes, but follow label directions. These fungicides are sold as solutions of potassium and/or sodium salts of phosphorous acid. To compare them, one should look at the phosphorous acid equivalent, which should be listed on the label.
Fungicide precautions. Phosphorous acid fungicide prices range from about $25 to $35 per gallon, and the application rate ranges from 2 to 5 pt/acre ($6.25 to $22 per acre, depending on the product and rate). Under high disease pressure, higher rates may need to be used and spray intervals tightened. These fungicides are formulated in salt form, so care must be taken not to exceed a certain concentration, as crop injury may result. In addition, if the concentration is too high, the pH may become so low that in tank mixes with copper products (particularly copper hydroxide such as Kocide), too much copper will become available and result in crop injury.

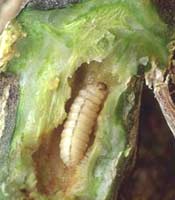 The squash vine borer, a key pest of winter squash, gourds and pumpkins in Kentucky is likely to become active in the next few weeks. Control for this insect pest is preventive where it has been a pest in the past as monitoring techniques are not very effective. For this reason, many growers usually notice the problem only after it has done its damage. Symptoms appear in mid-summer when a long runner or an entire plant wilts suddenly.
The squash vine borer, a key pest of winter squash, gourds and pumpkins in Kentucky is likely to become active in the next few weeks. Control for this insect pest is preventive where it has been a pest in the past as monitoring techniques are not very effective. For this reason, many growers usually notice the problem only after it has done its damage. Symptoms appear in mid-summer when a long runner or an entire plant wilts suddenly.
Infested vines usually die beyond the point of attack with squash vine borer. Sawdust-like frass near the base of the plant is the best evidence of squash vine borer activity. Careful examination will uncover yellow-brown excrement pushed out through holes in the side of the stem at the point of wilting. If the stem is split open, one to several borers are usually present. The caterpillars reach a length of 1 inch and have a brown head and a cream-colored body.
The adult squash vine borer is a stout dark gray moth with 'hairy' red hind legs, opaque front wings, and clear hind wings with dark veins. Unlike most moths, they fly about the plants during the daytime, appearing more like a paper wasp than a moth. Adult moths begin to emerge about the time the plants begin to run, and moth flight continues through mid August. The small brown eggs, laid individually on leaf stalks and vines, hatch in seven to 10 days. The newly hatched larva immediately bores into the stem. A larva feeds for 14 to 30 days before exiting the stem to pupate in the soil.
The key to squash vine borer management is controlling the borers before they enter the stem. Once inside the vine, insecticidal control is ineffective. Poor timing of sprays is the usual cause of inadequate control. Monitor plants weekly from mid-June through August for initial signs of the borer's frass at entrance holes in the stems. Very early signs of larval feeding indicate that other eggs will be hatching soon. Use two insecticide applications 7 days apart to control newly hatching larvae and continue to monitor for additional activity. Sprays need to penetrate the canopy to cover the vines to be effective.
Other Causes of Wilting. There can be several different causes of wilting of individual vines or the entire plant. It is important to determine what is the causing the problem so they proper preventive controls can be used next season. Often more than one factor may be causing a mixture of problems in a single field. Bacterial wilt transmitted by cucumber beetles will also begin to appear at this time. Single vines followed by the collapse of the entire plant is common. Prior to collapse, the plant usually retains its green color. Yellow vine decline, transmitted by squash bug, can also cause various types of wilts and decline. With some types of cucurbits, there will be yellowing of he plants before wilting.


 Adult Japanese beetles and masked chafers have begun to emerge. As is usually the case, it is difficult to predict how serious a problem these pests will be this year. Both Japanese beetles and masked chafers lay eggs in moist soil under turf. Their grubs then feed on turfgrass roots.
Adult Japanese beetles and masked chafers have begun to emerge. As is usually the case, it is difficult to predict how serious a problem these pests will be this year. Both Japanese beetles and masked chafers lay eggs in moist soil under turf. Their grubs then feed on turfgrass roots.
Japanese Beetles (Adults) - Detailed information on this pest can be found in ENT-5, Japanese Beetles in the Urban Landscape. Options for protecting landscape plants from foliage feeding adults are as follows:
Plant Selection- The best way to avoid perennial battles with adult Japanese beetles is to select plant material that is less preferred. Publication ENT-5 lists species and cultivars of trees and shrubs that are less likely to be attacked by beetles.
Hand Picking and Exclusion- For smaller plants, it may be practical simply to remove the beetles by hand. Volatile odors released from beetle-damaged leaves attract more beetles. By not allowing Japanese beetles to accumulate, plants will be less attractive to other beetles. One of the easiest ways to remove beetles from small plants is to shake them off early in the morning when the insects are sluggish. The beetles may be killed by shaking them into a bucket of soapy water. Highly valued plants such as roses can be protected by covering them with cheesecloth or other fine netting during peak beetle activity (usually late June to mid-July).
Insecticides- Carbaryl (Sevin) and several pyrethorid products such as bifenthrin (TalstarOne), cyfluthrin (Tempo, Bayer Advanced Lawn& Garden Multi-Insect Killer), deltamethrin (Deltaguard), lambda cyhalothrin (Scimitar, Spectracide Triazicide), and permethrin are labeled for control of adult Japanese beetles. The pyrethroids generally gave 2-3 weeks protection of plant foliage while carbaryl gave 1-2 weeks protection. Foliage and flowers should be thoroughly treated. The application may need to be repeated to prevent re-infestation during the adult flight period. Follow label directions and avoid spraying under windy conditions. For gardeners seeking a botanical alternative, Neem (e.g., Azatrol, Neem-Away from Gardens Alive), or Pyola (pyrethrins in canola oil) provided about 3-4 days deterrence of Japanese beetle feeding. Insecticidal soap, extracts of garlic, hot pepper, or orange peels, and companion planting, however, were found to be non-effective.
White Grubs - There is no reliable way to predict whether any given year will be a bad one for white grubs - the immature, turf-feeding stages of Japanese beetles, masked chafers, and certain other beetles. Moreover, since grub infestations tend to be localized and sporadic, only small percentages (< 10 percent) of Kentucky lawns require treatment, even in bad years for grubs.
Indicators of Infestation- White grubs and their resultant damage are not usually evident until August or September. Although sampling the turf is the only way to confirm that grubs are present, certain factors may indicate an increased risk of infestation later in the season. If your turf has a history of serious grub problems, there is a greater chance that adult beetles will return and re-infest the same areas. Sites with large numbers of adult beetles in June and July are more likely to have grubs in late summer. Early warning signs include swarms of brown, �-inch long masked chafer beetles skimming over the turf at dusk, or green June beetles buzz-bombing the turf by day in search of mates and egg-laying sites. Masked chafer and May beetle adults are also attracted to porch and streetlights at night. Heavy infestations of adult Japanese beetles feeding in the area might also foretell subsequent problems with grubs of that species.
Rainfall and soil moisture are critical factors affecting the extent of grub damage during a season. Frequent irrigation in June and July may attract egg-laying female beetles to the turf, especially if surrounding areas are dry. High soil moisture also increases egg survival. If lawns are irrigated during periods of dryness in June and July, be especially alert for signs of grubs later in the summer. Conversely, adequate soil moisture in August and September (when grubs are actively feeding) can help to hide root injury. Irrigated turf can sometimes tolerate 20 or more grubs per square foot before showing signs of injury.
Treatment Strategies - Two different strategies are available for controlling white grubs with insecticides: preventive and curative. Each approach has its own merits and limitations. With preventive control, the insecticide is applied as insurance, before a potential grub problem develops. Consequently, preventive control is best suited for high-risk sites with a history of grub problems, or turf sites where heavy beetle activity is noted.
Preventive control requires the use of insecticides with long residual activity in soil. Look for products containing the active ingredient imidacloprid (e.g. Merit�, Bayer Advanced� Season-Long Grub Control) or halofenozide (e.g. Mach 2�). Both of those ingredients have sufficient soil persistence to apply anytime from early June to mid-July and still control young grubs hatching from eggs from mid-July to early August. The optimum treatment period for these products is mid-June to mid-July.
Preventive treatments afford greater flexibility in application timing, and are easier to schedule and implement than are curative treatments. They often afford greater peace of mind to golf superintendents and lawn service companies because potential damage is avoided or minimized. The main drawback of preventive grub control is that the decision to treat must be made before knowing the extent of infestation. Grub outbreaks tend to be localized and sporadic and only a small percentage of lawns require treatment in a given year. Thus, preventive control often results in areas being treated unnecessarily. Good record keeping and observation will help in pinpointing grub-prone areas, which are the most logical candidates for preventive applications.
With curative control, treatment is applied in late summer - typically August or September - after the eggs have hatched and grubs are present. Ideally, the decision to treat is based on site inspection and sampling or past history of infestation. Since white grub infestations tend to be localized, the entire lawn often will not need to be treated. Grub "hot spots," which can be confirmed by sampling, are most likely to be full sun, south or west-facing slopes, lawns seeded with Kentucky bluegrass, lawns that were heavily irrigated during June and July, and turf areas that were damaged by grubs in previous years.
Proper timing of curative grub treatments can be tricky. Insecticides applied before early August may degrade before the eggs have hatched, whereas if the product is applied in late August or September, the grubs will be large and harder to kill and severe damage to turf may have already occurred. Granular formulations containing the active ingredient trichlorfon (e.g. Dylox, Bayer Advanced 24-hour Grub Control) are the fastest-acting, most effective insecticides for curative grub control. There is little benefit in applying a short-lived, curative-type product for white grubs in June or July.


 Discarded tire casings, and those used to weigh down tarps, will collect rainwater and can be excellent breeding sites for mosquitoes. A survey of mosquito larvae collected from "bunker tires" used to hold tarps over stored silage on New York dairy farms yielded over 13,000 larvae representing 12 mosquito species. All but two of them could be important vectors of viruses that could affect humans and livestock.
Discarded tire casings, and those used to weigh down tarps, will collect rainwater and can be excellent breeding sites for mosquitoes. A survey of mosquito larvae collected from "bunker tires" used to hold tarps over stored silage on New York dairy farms yielded over 13,000 larvae representing 12 mosquito species. All but two of them could be important vectors of viruses that could affect humans and livestock.
Information on reducing mosquito breeding sites when using waste tires as anchors or alternatives is available at
http://cropsoil.psu.edu/People/faculty/PDF/Reducing_mosquito_sites%20.pdf.


During the past week, field crop samples diagnosed included zinc and potassium deficiency, and Pythium and Rhizoctonia root rots on corn; boron deficiency on alfalfa; Phytophthora root rot on alfalfa; Pythium and Rhizoctonia root rots, and potassium deficiency on soybean; Pythium root rot on (sweet) sorghum; bacterial blackleg, Pythium root rot, black root rot, tomato spotted wilt virus and temporary phosphorus deficiency on tobacco.
On fruit and vegetable samples, we diagnosed transplant shock, iron deficiency and Botrytis twig blight on blueberry; Botrytis gray mold and Phomopsis leaf blight on strawberry; fire blight and cedar-apple rust on apple; bacteria spot and scab on peach; tomato spotted wilt virus on pepper; and Sclerotinia stem rot and bacterial canker on tomato.
On ornamentals, we diagnosed Botrytis blight on pansy; bacterial soft rot on dahlia; Pythium root rot and impatiens necrotic spot virus on impatiens; Ascochyta dieback on clematis; Kabatina twig blight on juniper; powdery mildew, thrips injury; rose rosette and rose mosaic virus on rose; Volutella canker on boxwood; anthracnose on maple and oak; Verticillium wilt on redbud; Phyllosticta leaf spot (and blight) on witchhazel; and downy spot (Microstroma) on walnut.


UKREC-Princeton, KY, June 3 - 10, 2005| Black Cutworm
| 8
| True Armyworm
| 10
| Corn Earworm
| 5
| European Corn Borer
| 0
| Southwestern Corn Borer
| 49
| Fall Armyworm
| -
| | |
Hardin Co., KY, June 3 - 10, 2005| Black Cutworm
| -
| True Armyworm
| -
| Corn Earworm
| -
| European Corn Borer
| 7
| Southwestern Corn Borer
| 42
| Fall Armyworm
| 2
| | |
View Princeton trap counts for the entire 2005 season at - http://www.uky.edu/Ag/IPMPrinceton/Counts/2005trapsfp.htm
Fulton County trap counts are available at -http://ces.ca.uky.edu/fulton/anr/Insect%20Counts.htm
For information on trap counts in southern Illinois visit the Hines Report at - http://www.ipm.uiuc.edu/pubs/hines_report/comments.html The Hines Report is posted weekly by Ron Hines, Senior Research Specialist, at the University of Illinois Dixon Springs Agricultural Center.
NOTE: Trade names are used to simplify the information presented in this newsletter. No endorsement by the Cooperative Extension Service is intended, nor is criticism implied of similar products that are not named.
Lee Townsend
Extension Entomologist