

The Kentucky Geological Survey has archived this material, meaning (1) it is for reference, research, or recordkeeping; (2) it was created before April 24, 2026; (3) and the material has not been changed or altered since being archived. Please refer to our KGS Accessibility page for more information.
KGS Home > Geology of Kentucky
How Geodes Form
The concentration of geodes in the Lower to Middle Mississippian rocks of Kentucky, Indiana, Illinois, and Tennessee is a result of both large-scale depositional and burial processes and natural alteration of the original limy sediments through mineral-rich fluids, termed dolomitization. Dolomitization is a process by which limestone is converted to dolomite.
Figure 2 is a graphic representation of how void spaces in a sedimentary rock layer are formed and then filled with minerals to form geodes, solid mineral nodules, and agates. Voids can be filled in several ways (multiple “paths” in the diagram). Most commonly, voids are completely filled with the mineral quartz, but the filling can be by a wide variety of minerals.
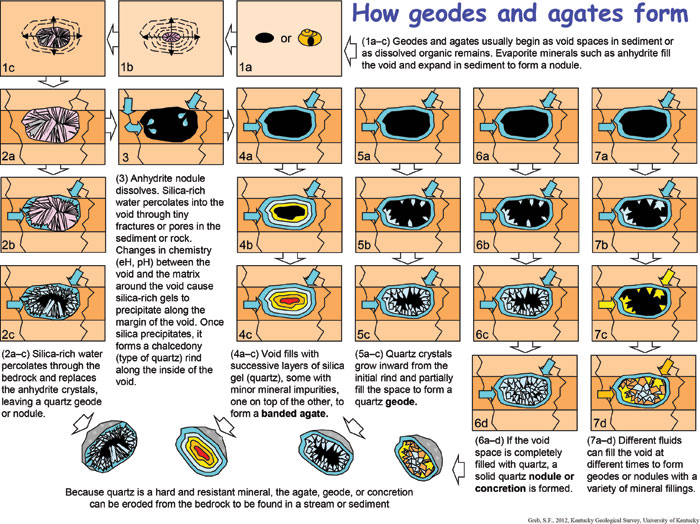
Figure 2. How geodes, solid mineral nodules, and agates form. Select here for a pdf version.
1. Dolomitization and anhydrite.
Explanations of the geode-forming process by Chowns and Elkins (1974) Miliken (1979), Maliva (1987), and Barwood and Shaffer (no date) are in general agreement that Fort Payne geodes began as the mineral anhydrite (CaSO4) (Fig. 2), although opinions differ as to when the anhydrite formed and the water depths in which the fluids that led to mineral precipitation formed. Most studies are in general agreement that the migration of mineral-rich, dolomitizing fluids through the Fort Payne, Borden, and Salem-Warsaw sediments contributed to the formation of anhydrite nodules. Dolomitizing fluids are generally magnesium- and sulfate-rich fluids. Sulfates are compounds which that contain sulfur and oxygen as SO42. Anhydrite and gypsum are common sulfate minerals associated with dolomitization.
Some geodes have needle-shaped crystals which are more similar in shape to anhydrite than quartz, although they are now composed of the mineral quartz (Fig. 3A). When the original shape of a precursor mineral crystal is preserved in the existing crystal it is termed a pseudomorph. Some pseudonodules (incipient geodes) on bedding surfaces of the Fort Payne Formation also retain the general appearance of small anhydrite nodules, although they are now quartz nodules (Fig. 3B). Other geodes appear to fill vertical cracks in the bedrock (Fig. 3C), some of which follow linear trends on bedding surfaces (Fig. 3D). These attest to the migration of mineralizing fluids through cracks in the rock. Still other nodules and geodes appear to have replaced fossil skeletal material or are concentrated along burrow horizons. Organic material and voids within fossil skeletal material and burrows likely were common points of nucleation for anhydrite minerals (Maliva, 1987).

Figure 3. Quartz nodules and geodes in bedrock along Lake Cumberland.
(A) Geode with needle-shaped quartz crystals, which are possibly pseudomorphs after anhydrite.
(B) Small incipient cauliflower-shaped quartz nodules on a bedding surface, which are similar in shape to anhydrite nodules.
(C) Geodes filling a vertical, wedge-form void in a bed, similar to a fracture fill. Pen is approximately 5 in. long.
(D) Bedding-plane surface with geodes arranged in linear orientations parallel to the long axis of the hammer (white arrows).
Expansion of void-filling or replacement anhydrite into circular to elliptical nodules must have occurred while the surrounding sediment was still relatively soft and pliable. In some cases, layering and bedding around geodes in bedrock appear disrupted or contorted around the geode. When new, deep road cuts in the Fort Payne, Warsaw, or Borden Formations are excavated, small masses of anhydrite of anhydrite are sometimes encountered in the unweathered bedrock, which dissolve, and leave rims of silica around the resulting void. Downhole borehole cameras in wells into the Fort Payne and Warsaw around Bowling Green, Ky., have also encountered thin beds and nodules of anhydrite (personal communication, Randy Shields, 2007, oil and gas driller).
2. Silica sources.
An essential requirement for geodes is a source of easily dissolved silica to make the quartz filling. Although sediment (silt, sand, etc.) generally contains large amounts of quartz (SiO2), the silica in those sediment grains is not easily dissolved into solutions (non-soluble) where it is available to be precipitated again. For the Fort Payne Formation, a more available and more easily mobilized source of silica was biogenic silica. Biogenic silica is hydrated, amorphous silica, which is extracted from marine water and biochemically converted into silica structures by organisms such as sponges and radiolarians. Radiolarians are tiny protozoan zooplanktons, which build their skeleton (called tests) from amorphous silica (Fig. 4). Similarly, demosponges and hexactinellid (glass) sponges biochemically convert amorphous silica into skeletal structures called spicules (Fig. 4).
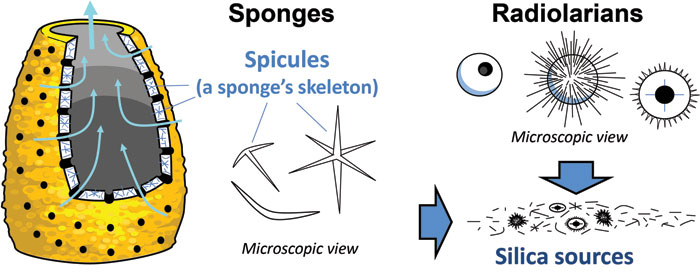
Figure 4. Sponges and radiolarians naturally use silica from nature to make their skeletons.
The Fort Payne Formation was originally deposited in a shallow marine sea, on a ramp or slope offshore of the Borden delta complex. During this part of the Mississippian Period (Osagean stage), deeper and colder water filled the Ouachita Trough, in what is now Arkansas (Fig. 5). Cold waters from the trough would have flowed up the depositional slope into what is now Kentucky. Upwelling of cold, nutrient-rich water against the marine ramp or slope from deeper waters to the west provided ideal conditions for siliceous sponges to flourish (see for example, Lowe, 1975). The Fort Payne Formation is partly equivalent to the Arkansas Novaculite in the Ouachita Trough. Novaculites are highly siliceous, bedded chert deposits. The source for much of the silica in the novaculites has been attributed to biogenic silica from sponges and radiolarians (Parke and Croneis, 1969; Lowe, 1975; Jones and Knauth, 1979). When the radiolarian or sponge dies, the silica tests and spicules are all that remain of the organisms. The silica in spicules from dead sponges forms much of the fine-grained matrix of the shaly parts of the Fort Payne Formation and may have been the source for silica to form geodes.
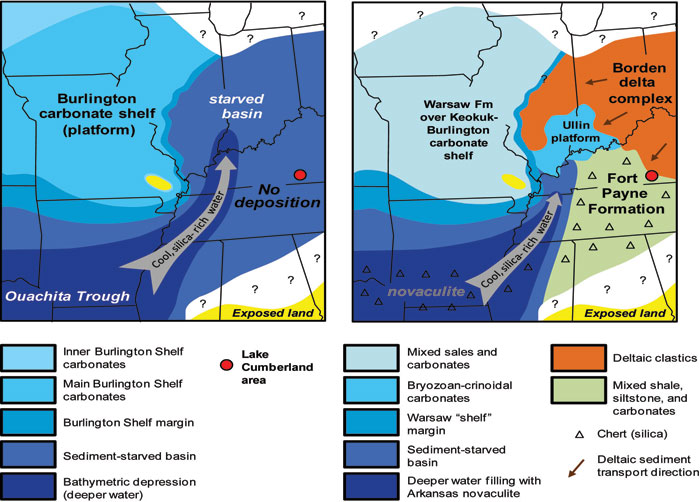
Figure 5. Regional paleogeographic setting of the Fort Payne Formation (from Lasemi and others, 2003, Fig. 11,
included here with permission of authors and Society for Sedimentary Geology).
Sponges and radiolarians in deeper water to the west, were likely the source for biogenic silica.
3. Silicification.
Silicification is the process of introducing silica (Si) into a sediment or fossil fragment and replacing the original material or filling void spaces in the material with silica. The most common mineral formed through silicification is quartz (SiO2). The dolomitizing fluids that moved through the Fort Payne, Borden, and Salem-Warsaw Formations were acidic. These fluids dissolved sponge spicules and mobilized (put into solution) amorphous silica into the fluids. Silica gels formed in pore spaces and as replacement minerals for anhydrite (Fig. 2). Replacement of anhydrite by silica gels along the contact with the surrounding sediment created a quartz rind (the outer surface of a nodule or geode). Petrographic work (Chowns and Elkins, 1974; Barwood and Shaffer, no date) indicates anhydrite inclusions are common within the microcrystalline parts of rinds in quartz geodes.
Silicification proceeded from the outer rind toward the center of the anhydrite nodules (Maliva, 1987). Quartz nodules were formed when all of the anhydrite was dissolved and the entire void space was filled by crystalline quartz (solid nodule in Fig. 2). In other areas, continued precipitation of silica gels with minor mineral impurities resulted in banded, agate-filled nodules (Fig. 2). Agate is Kentucky’s official State Gemstone, although it is a mineral (a variety of the mineral quartz). In geodes, only part of the void is filled, and the interior tends to be lined with quartz crystals (Fig. 2), although other minerals also occur.
In some cases, skeletal material (crinoids, brachiopods, gastropods) were also replaced by silica. Crinoids and gastropods make their skeletons and shells from the mineral aragonite, a variety of calcite. In some cases, original anhydrite may have nucleated within shells, and then expanded, ultimately breaking and expanding the shells (Maliva, 1987). Later, silica replacement increased or expanded the original size of the skeletal material even further. Hence, the silica-replaced after anhydrite-replaced fossils look like giant versions of their smaller (calcite-cemented) cousins. Similar to mineral replacement, when the original shape of a fossil is preserved in quartz it is termed a pseudomorph. In most cases, expansion results in loss of much of the original structure of the fossil material. In some cases, however, partial preservation of original animal structures or arrangements of structures can be determined in the expanded concretion or geode, which allow interpretation of the original fossil. Expanded brachiopods, which still retain a hingeline have been reported. More common is the preservation of pentagonal plate shapes or vertical changes in plate shape and sizes reminiscent of the structural arrangements of plates in crinoids. Quartz, when not precipitated as a pseudomorph, would more commonly produce hexagonal (6-sided) crystals and shapes.
4. Sulfate reduction.
Although most nodules and concretions were completely replaced and filled with quartz, and sharp-faced (euhedral), hexagonal quartz crystals are common fills in Fort Payne, Borden, and Salem-Warsaw geodes, a suite of accessory minerals, including barite, calcite, pyrite, sphalerite, native sulfur, and a variety of iron minerals is also found within nodules, concretions, and geodes (Figs. 2, 6). Barite (BaSO4), like anhydrite, is a sulfate mineral and likely precipitated with anhydrite. In contrast, pyrite (FeS2) and sphalerite (ZnS) are sulfide minerals. In sulfates, sulfur (S) is bound with oxygen (SO4). In sulfides, the sulfur atom has two negative charges (S2-) and binds with elements other than oxygen. Sulfide mineral precipitation and the precipitation of native sulfur (S2-), likely occurred as a result of sulfate reduction in dolomitizing fluids (Chowns and Elkins, 1974; Barwood and Shaffer, no date). Sulfate reduction is a process through which the oxidation state of sulfate compounds or minerals is decreased (or the number of electrons is increased). Sulfate reduction can also increase pH (acidity) in pore fluids. In some cases, this may have removed sulfate-mineral cores within the center of nodules to generate a void space and form a geode.

Figure 6. Geode from the Fort Payne Formation at Lake Cumberland containing iron minerals.
Where Did the Dolomitizing Fluids Come From?
Dolomitizing fluids and anhydrite precipitation can occur in many ways, at many depths, and at many times relative to original deposition of sediments. The following are some of the models proposed for the origin of dolomitizing fluids in the Borden, Fort Payne, and Salem-Warsaw-Formations.
Shallow water: Modern anhydrite nodules and dolomite have been documented forming in arid, hypersaline coastlines (called sabkhas or sahbkas) in the Persian Gulf. In arid climates, evaporation concentrates salts in shallow surface waters (ponds, lagoons) and near-surface groundwater creating dense, hypersaline fluids. Evaporation on supratidal flats can also pull sea water and pore fluids through the coastal sabkha sediments, leading to the precipitation of evaporate minerals, including anhydrite and gypsum, at the surface, or in near-surface sediments. Primary dolomite can also be precipitated in supratidal flats, possibly due to reactions with microbrial mats on the tidal flats (Wells, 1962; McKenzie et al., 1980; Patterson and Kinsman, 1982).
Magnesium-rich waters can also into seep into the near subsurface to form dolomitizing fluids and secondary dolomite (Adams and Rhodes, 1960; DeFeyes and others, 1965; Shinn, 1983). This is termed seepage reflux. In reflux, movement of supersaturated groundwater is downward and seaward because the waters are dense and displace non-saline or normal-saline pore waters in the sediments. Numerous studies have used these general models to infer that the dolomitizing fluids which led to geode formation in the Fort Payne, Borden, and Salem-Warsaw Formations originated in shallower, Mississippian-age carbonates. Here are some summaries:
Maliva (1987) inferred that the origin of dolomitizing fluids in the Fort Payne was in shallow, hypersaline waters and sediments of the Middle Mississippian St. Louis Limestone. Rather than primary precipitation of super-saturated groundwater in St. Louis sabkhas, Maliva (1987) proposed seepage reflux of lagoonal St. Louis Limestone waters down through the Salem-Warsaw sediments. Saturated waters moved seaward and downward, increasing concentrations of calcium and other minerals along the way. In the Harrodsburg Limestone and Fort Payne Formation (equivalent to part of the Ramp Creek Formation in Indiana), these fluids became supersaturated with respect to anhydrite.
Barwood and Schaffer (no date) advocated Maliva’s model, and noted that anoxic conditions basinward, at deeper depths, may have contributed to some of the mineral reactions that led to geode formation.
Deep water. More recently, research has shown that diagenetic dolomites can also be formed through the migration of fluids originating in deeper waters. Lumsden (1988; 2003) proposed the possibility of deep-water microbial sulfate reduction as a source of dolomitizing fluids, which is an attractive alternative to the shallow-water origin because of the deeper water environments proposed for Fort Payne deposition.
Back to Geodes in the Fort Payne, Borden, and Salem-Warsaw Formations
Back to top of Geology of the Fort Payne Formation